UNITED STATES
SECURITIES AND EXCHANGE COMMISSION
Washington, D.C. 20549
FORM 6-K
REPORT OF FOREIGN PRIVATE ISSUER
PURSUANT TO RULE 13a-16 OR 15d-16
UNDER THE SECURITIES EXCHANGE ACT OF 1934
Date of report: September 22, 2025
Commission File Number: 001-38844
GENFIT S.A.
(Translation of registrant’s name into English)
Parc Eurasanté
885, avenue Eugène Avinée
59120 Loos, France
(Address of principal executive office)
Indicate by check mark whether the registrant files or will file annual reports under cover of Form 20-F or Form 40-F:
☒ Form 20-F ☐ Form 40-F
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(1): ☐
Indicate by check mark if the registrant is submitting the Form 6-K in paper as permitted by Regulation S-T Rule 101(b)(7): ☐
INCORPORATION BY REFERENCE
The contents of this report on Form 6-K (including Exhibit 99.1) are hereby incorporated by reference into the registrant’s registration statement on Form F-3 (File No. 333-271312) and registration statement on Form S-8 (File No. 333-271311) and related prospectuses, as such registration statements and prospectuses may be amended from time to time, and to be a part thereof from the date on which this report is filed, to the extent not superseded by documents or reports subsequently filed or furnished. Information contained on, or that can be accessed through, any website included in Exhibit 99.1 is expressly not incorporated by reference.
EXHIBIT LIST
| Exhibit | Description | |||||||||||||||||||||||||||||||||||||||||||||||||
| 99.1 | ||||||||||||||||||||||||||||||||||||||||||||||||||
SIGNATURES
Pursuant to the requirements of the Securities Exchange Act of 1934, the registrant has duly caused this report to be signed on its behalf by the undersigned, thereunto duly authorized.
| GENFIT S.A. | ||||||||||||||||||||
| Date: September 22, 2025 | By: | /s/ Pascal PRIGENT | ||||||||||||||||||
| Name: Pascal PRIGENT | ||||||||||||||||||||
| Title: Chief Executive Officer | ||||||||||||||||||||
| Half-Year Business and Financial Report | |||||
at June 30, 2025 | |||||
 | |||||
| Table of Contents | |||||||||||
| 1. | OVERVIEW OF THE GROUP AND ITS MAIN R&D PROGRAMS | ||||||||||
| 2. | HALF-YEAR MANAGEMENT REPORT | ||||||||||
2.1 | Key Events of the First Half of 2025 and Main Events after the Reporting Period | ||||||||||
2.2 | Strategy and Outlook | ||||||||||
2.3 | Operating and Financial Review | ||||||||||
2.4 | Main Transactions with Related Parties | ||||||||||
2.5 | Main Risks and Uncertainties | ||||||||||
| 3. | HALF-YEAR CONDENSED CONSOLIDATED FINANCIAL STATEMENTS AT JUNE 30, 2025 | ||||||||||
3.1 | Consolidated Statements of Financial Position | ||||||||||
3.2 | Consolidated Statements of Operations | ||||||||||
3.3 | Consolidated Statements of Other Comprehensive Loss | ||||||||||
3.4 | Consolidated Statements of Cash Flows | ||||||||||
3.5 | Consolidated Statements of Changes in Equity | ||||||||||
3.6 | Notes to the Consolidated Financial Statements | ||||||||||
| 4. | STATUTORY AUDITORS’ LIMITED REVIEW REPORT ON 2025 HALF-YEAR CONDENSED CONSOLIDATED FINANCIAL STATEMENTS | ||||||||||
| 5. | DECLARATION BY THE PERSON RESPONSIBLE FOR THE INFORMATION | ||||||||||
Disclaimer | ||
This report contains certain forward-looking statements, including those within the meaning of the Private Securities Litigation Reform Act of 1995 with respect to GENFIT, including, but not limited to statements about GENFIT’s corporate strategy and objectives, our achievement of key milestones enabling us to receive payments under our license agreements, the potential of Iqirvo® (elafibranor) to receive marketing authorization and successful launch and commercialization in countries other than those in which it is currently approved and commercialized and/or in indications other than PBC, our achievement of the necessary objectives to obtain the future €55 million in additional payments under the royalty financing agreement signed with HCRx (Royalty Financing), anticipated timing for study enrollment and data readouts, in particular regarding our development programs for G1090N in the prevention and/or treatment of ACLF and for GNS561 in CCA, and development plans for our other pipeline programs, in particular those related to SRT-015, CLM-022 and VS-02 HE in ACLF, and VS-01 in UCD, the expected timing for potential regulatory approvals and the impact of the development of our programs and our internal organization, our ability to qualify for and obtain specific regulatory pathways, as well as our financial outlook including cash flow and cash burn projections as updated following the termination of our VS-01 in ACLF research program and business and R&D activity projections for 2025 and beyond. The use of certain words, including “believe, “potential”, “expect”, “target”, “may”, “should” and “will” and similar expressions, is intended to identify forward-looking statements. Although the Company believes its expectations are based on the current expectations and reasonable assumptions of the Company’s management, these forward-looking statements are subject to numerous known and unknown risks and uncertainties, which could cause actual results to differ materially from those expressed in, or implied or projected by, the forward-looking statements. These risks and uncertainties include, among other things, the uncertainties inherent in research and development, including in relation to safety of drug candidates, cost of, progression of, and results from, our ongoing and planned clinical trials, review and approvals by regulatory authorities in the United States, Europe and worldwide, of our drug and diagnostic candidates, potential commercial success of Iqirvo® (elafibranor), exchange rate fluctuations, our continued ability to raise capital to fund our development, as well as those risks and uncertainties discussed or identified in the Company’s public filings with the AMF, including those listed in Chapter 2 “Risk Factors and Internal Control” of the Company’s 2024 Universal Registration Document filed with the AMF on April 29, 2025, which is available on the Company’s website (www.genfit.com) and on the website of the AMF (www.amf-france.org) and public filings and reports filed with the U.S. Securities and Exchange Commission (“SEC”) including the Company’s 2024 Annual Report on Form 20-F filed with the SEC on April 29, 2025 and subsequent filings and reports filed with the AMF or SEC, including this Half-Year Business and Financial Report at June 30, 2025 or otherwise made public, by the Company.
In addition, even if the Company’s results, performance, financial condition and liquidity, and the development of the industry in which it operates are consistent with such forward-looking statements, they may not be predictive of results or developments in future periods.
These forward-looking statements speak only as of the date of publication of this document. Other than as required by applicable law, the Company does not undertake any obligation to update or revise any forward-looking information or statements, whether as a result of new information, future events or otherwise.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
1 | ||||||||
| 1. | OVERVIEW OF THE GROUP AND ITS MAIN R&D PROGRAMS | |||||||
About GENFIT
GENFIT is a biopharmaceutical group (the "Group” or “GENFIT” or the "Company”) committed to improving the lives of patients with liver diseases whose medical needs remain largely unmet, with particular attention to rare and life-threatening diseases including their acute manifestations. The Group includes the parent company GENFIT SA, founded in 1999 incorporated under French law and two wholly-owned subsidiaries: GENFIT Corp. (American subsidiary) and Versantis AG (Swiss subsidiary) whose accounts are consolidated with those of GENFIT SA.
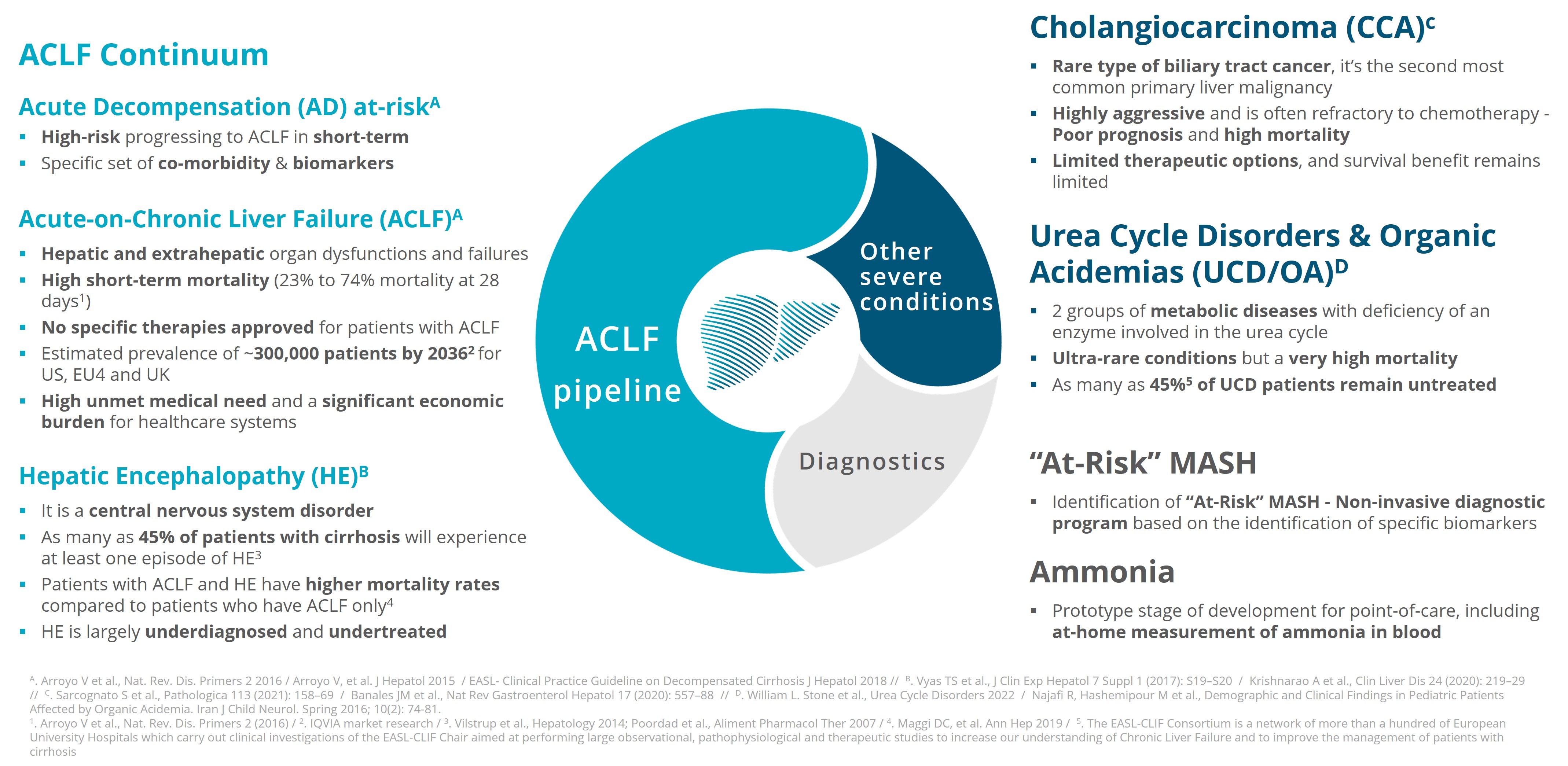
GENFIT is a pioneer in liver disease research and development with a rich history and a solid scientific heritage spanning more than two decades. Today, GENFIT has built
up a diversified and rapidly expanding R&D portfolio of programs at various stages of development. The Company focuses on Acute-on-Chronic Liver Failure (ACLF). Its ACLF franchise includes four assets under development: G1090N, SRT-015, CLM-022 and VS-02-HE, based on complementary mechanisms of action using different routes of administration. Other assets target other serious diseases, such as GNS561 in Cholangiocarcinoma (CCA), and VS-01-HAC in Urea Cycle Disorders (UCD) and Organic Acidemias (OA).
GENFIT's expertise in the development of high-potential molecules from early to advanced stages, and in pre-commercialization, was demonstrated in the accelerated approval of Iqirvo® (elafibranor)1 by the U.S. Food and Drug Administration, the European Medicines Agency and the Medicines and Healthcare Regulatory Agency in the UK for Primary Biliary Cholangitis (PBC).
Beyond therapies, GENFIT also has a diagnostic franchise including NIS2+® in Metabolic dysfunction-Associated Steatohepatitis (MASH, formerly known as NASH for non-alcoholic steatohepatitis) and TS-01 focusing on blood ammonia levels.
GENFIT is headquartered in Lille, France and has offices in Paris (France), Zurich (Switzerland) and Cambridge, MA (USA). The Company is listed on the Nasdaq Global Select Market and on the Euronext regulated market in Paris, Compartment B (Nasdaq and Euronext: GNFT). In 2021, Ipsen became one of GENFIT's largest shareholders, acquiring an 8% stake in the Company's capital. www.genfit.com
Overview of the main R&D programs of the Company
GENFIT remains faithful to its vocation and its specialization in hepatology, and is evolving towards having a portfolio that covers several serious and rare liver diseases that are characterized by largely unmet medical needs and their significant impact on patients’ lives.

1 Elafibranor is marketed and commercialized, notably in the U.S and Europe, by Ipsen under the trademark Iqirvo®.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
2 | ||||||||
Over the past several years, GENFIT has made a strategic pivot towards Acute-On-Chronic Liver Failure (ACLF) — covering a broad spectrum of conditions across a disease continuum including Acute Decompensation (AD) of liver cirrhosis, Hepatic Encephalopathy (HE), etc. — and other life-threatening liver conditions, broadening its research pipeline to include promising drug candidates that aim to meet the urgent and unmet needs of this challenging condition:

Upcoming milestones, data announcements and launch dates are anticipated and subject to change. ACLF: Acute-on-Chronic Liver Failure. CCA: Cholangiocarcinoma; HAC: Hyperammonemic Crises; UCD = Urea Cycle Disorders ; OA = Organic Acidemias ; HE: Hepatic Encephalopathy; MASH: Metabolic dysfunction-Associated Steatohepatitis; G1090N Repositioned (Nitazoxanide or NTZ) and reformulated molecule.
All drugs under development are investigational compounds that have not been reviewed nor been approved by a regulatory authority in targeted indications. GENFIT has licensed the exclusive worldwide rights of ASK1 Inhibitor SRT-015 (injectable formulation in acute liver disease) from Seal Rock Therapeutics. GENFIT licensed the exclusive worldwide rights of CLM-022, a potential first-in-class inflammasome inhibitor, from Celloram Inc. GENFIT purchased the intellectual property rights underlying GNS651 from Genoscience Pharma in January 2025. Labcorp has a non-exclusive license for the development and commercialization of NIS4® technology to power a next-generation MASH diagnostic laboratory-developed test (LDT) to identify patients with at-risk MASH in the United States and Canada. NIS2+® is a next-generation technology derived from NIS4®.
In December 2021, we entered into a license agreement with Ipsen, granting Ipsen a global license2 to develop, manufacture, and commercialize elafibranor for the treatment of patients with Primary Biliary Cholangitis (PBC) and other indications. GENFIT led the research and clinical development of elafibranor through the Phase 3 ELATIVE® trial in PBC. Ipsen is now responsible for all future clinical development, including completion of the long-term extension of the Phase 3 ELATIVE® trial, as well as global commercialization which notably began in several countries including the United States and several European Economic Area (EEA) countries in 2024 under the name Iqirvo® (elafibranor). GENFIT has received and continues to receive revenue from this agreement in the form of milestone payments and royalties on sales subject to thresholds. For more information regarding the license agreement with Ipsen, see Note 24. Under the royalty financing agreement signed with HCRx in March 2025, HCRx receives a portion of royalties on sales of Iqirvo® (elafibranor) payable to GENFIT under its licensing agreement with Ipsen, up to an agreed upon cap after which all future royalties will revert back to GENFIT (see below).
2 Except for China, Hong Kong, Taiwan, and Macau.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
3 | ||||||||
| 2. | HALF-YEAR MANAGEMENT REPORT | |||||||
| 2.1 | Key Events of the First Half of 2025 and Main Events after the Reporting Period | |||||||
Financing: Closing of a royalty financing agreement with HCRx and near-total reimbursement of convertible debt, significantly extends the cash runway of the Company | ||||||||
On March 20, 2025, GENFIT announced the closing of a royalty financing transaction (Royalty Financing) with HealthCare Royalty (HCRx) providing up to €185 million non-dilutive capital: €130 million upfront, with eligibility to receive up to €55 million in two additional installments based on near-term sales milestones for Iqirvo® (elafibranor), and can be exercised at the discretion of GENFIT upon achievement of such milestones. In return, HCRx will receive a portion of royalties on global3 sales of Iqirvo® (elafibranor) payable to GENFIT under its licensing agreement with Ipsen, up to an agreed upon cap after which all future royalties will revert back to GENFIT.
GENFIT retains rights to all future regulatory, commercial and sales-based milestone payments from Ipsen under the Ipsen agreement.
The Royalty Financing has significantly extended GENFIT’s cash runway, beyond the end of 2028, enabling the Company to further develop its pipeline focused on Acute-on-Chronic Liver Failure (ACLF) and support general corporate purposes. This estimation is based on current assumptions and programs and does not include exceptional events. This estimation assumes i) our expectation to receive significant future commercial milestone revenue pursuant to the license agreement with Ipsen and Ipsen meeting its sales-based thresholds4, ii) drawing down all additional installments under the Royalty Financing, and iii) the reimbursement at maturity in October 2025 of any OCEANEs not converted or repurchased and cancelled, and iv) the discontinuation of the VS-01 program in ACLF as outlined below at the end of this section.
Concurrently with the Royalty Financing, GENFIT proposed to OCEANEs holders the possibility to enter into a put option agreement for the repurchase of their OCEANEs (Repurchase). Following this proposal, holders of OCEANEs exercised their put option for a total of 1,882,891 OCEANEs, i.e. 99% of the total number of OCEANEs outstanding. At a price of €32.75 per bond, this represented a total Repurchase amount of €61.7 million.
For more information regarding the Royalty Financing transaction, see Note 2.1 to the financial statements for the half year ended June 30, 2025.
PBC: New milestone payment, encouraging information regarding sales of Iqirvo® (elafibranor) by our partner Ipsen, and withdrawal of market competitor in US Market | ||||||||
€26.5 million milestone payment following the approval of pricing and reimbursement of Ipsen’s Iqirvo® (elafibranor) in Italy | ||
In May 2025, Iqirvo® (elafibranor) was granted pricing and reimbursement in Italy for PBC. This major step unlocked a new milestone payment of €26.5 million under our licensing and collaboration agreement with Ipsen, due upon pricing and reimbursement of Iqirvo® (elafibranor) in three major European markets5.
Accelerated sales growth for Iqirvo® (elafibranor) in the first half of 2025 and withdrawal of OCALIVA® (obeticholic acid) for PBC from the US Market | ||
In July, 2025, Ipsen reported “accelerated sales growth of €59 million in the first half of 2025 in the U.S. and in Europe (mainly Germany & U.K.) driven by increasing uptake from new patients, switch and market expansion sales for Iqirvo® (elafibranor)6“.
3 Excluding China, Hong Kong, Taiwan and Macau.
4 For a total of €586 thousand as of the date of this report.
5 Iqirvo® (elafibranor) has already been granted pricing and reimbursement in the UK and in Germany in 2024.
6 https://www.ipsen.com/websites/ipsen_com_v2/wp-content/uploads/2025/07/31081007/H1-2025-results-presentation.pdf
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
4 | ||||||||
In September, 2025, Intercept announced voluntary withdrawal of OCALIVA® (obeticholic acid) for PBC from the US Market7.
PSC: Positive late-breaking Phase 2 data for elafibranor presented by Ipsen at EASL Congress 2025 | ||||||||
In May 2025, Ipsen presented data from its late-breaking abstract on elafibranor, highlighting favorable safety profile and significant efficacy in Primary Sclerosing Cholangitis (PSC), at the European Association for the Study of the Liver (EASL).
Efficacy results of Ipsen’s Phase 2 ELMWOOD trial (LB25222/OS089) showed that patients on elafibranor had significant dose-dependent reductions in alkaline phosphatase (ALP), with patients on elafibranor 80 mg and 120 mg having significant reductions at week 12 versus placebo (−103.2 U/L and −171.1 U/L vs +32.1 U/L; p < 0.0001), and improvements observed as early as week 4. Similar findings were seen in other biochemical liver parameters, including alanine aminotransferase (ALT) and gamma-glutamyl transferase (GGT), which are important biochemical markers of disease progression. Patients on elafibranor also had stabilization in Enhanced Liver Fibrosis (ELF), a non-invasive marker of liver fibrosis, versus patients on placebo at week 12. Additionally, patients on elafibranor 120 mg experienced improvements in pruritus compared with patients on placebo according to the Worst Itch Numeric Rating Scale (WI NRS) score (-0.96 vs -0.28; p<0.05).8
Should elafibranor be approved in a second indication after PBC, GENFIT would also be eligible to milestone payments and royalties under the licensing and collaboration agreement with Ipsen .
ACLF: Discontinuation of VS-01 program, refocused on UCD | ||||||||
On September 19, 2025, GENFIT announced its decision to discontinue its VS-01 program in ACLF (Acute-on-Chronic Liver Failure), and reprioritize its development on UCD (Urea Cycle Disorder):
•GENFIT’s decision followed the occurrence of a peritonitis case reported as Serious Adverse Event (SAE) in the UNVEIL-IT® clinical trial evaluating VS-01 in patients with ACLF grade 1, 2 or 3a and ascites and subsequent review and feedback from the independent Data Monitoring Committee (iDMC). The committee concluded that the trial could continue but required additional data and monitoring. Despite the possibility to move ahead with the study, GENFIT decided – after considering the target population’s clinical profile as well as the implications of this type of safety signals for the benefit/risk ratio of VS-01 in this indication – to discontinue both UNVEIL-IT® and the proof-of-concept study evaluating VS-01 in patients with Hepatic Encephalopathy (HE) grades 2 to 4 in the presence of Acute Decompensation (AD) or ACLF grade 1 and ascites.
•GENFIT will continue the preclinical evaluation of VS-01 in UCD, a genetically driven disorder characterized by Acute Hyperammonemic Crisis (HAC). The condition, patients and drug administration set-up will be very different from what they were in ACLF. There is a significant unmet medical need in this indication, and based on ammonia clearance data, we believe VS-01 has the potential to be a useful therapeutic option for children affected by this disease.
•GENFIT remains fully committed to ACLF and associated conditions such as Acute Decompensation (AD) or Hepatic Encephalopathy (HE). ACLF is characterized by a critical unmet medical need, with no approved treatment options for patients facing poor prognosis and life-threatening risks. Since we embarked in this therapeutic area, we have engaged in multiple KOL interactions and observed growing interest in this indication, together with clear support for our clinical strategy. This feedback reinforces our confidence in our plan and validates our positioning. In this context, we ambition to accelerate the development of the four other assets currently under development in ACLF, which are all based on different mechanisms of action and use different routes of administration. We hope to deliver positive results, as we move forward, starting with safety data and markers of efficacy on healthy volunteers with G1090N, expected at the end of this year. Other programs in the ACLF pipeline are SRT-015, CLM-022 and VS-02-HE.
Earlier in May 2025, GENFIT participated at the European Association for the Study of the Liver (EASL) International Congress 2025 with several posters presenting its latest progress in ACLF. The congress highlighted the growing importance of ACLF within the hepatology community.
1.Posters presented by GENFIT at EASL Congress
Six posters have been presented at EASL Congress mainly covering several of GENFIT’s assets positioned in ACLF and GENFIT’s latest work on Real-World data in ACLF.
Among posters related to our programs:
•Poster #1: NTZ alleviates stress-induced hepatocyte cell death through modulation of oxidative stress and DNA damage signaling pathways in ACLF models
•Poster #2: Efficacy of the apoptosis-signal-regulating kinase 1 (ASK1) inhibitor SRT-015 in vivo and in vitro pathogen-associated molecular patterns (PAMPs)-induced disease models
7 https://www.interceptpharma.com/about-us/news/?id=3148535
8 Levy. C. et al. Elafibranor for Primary Sclerosing Cholangitis: The ELMWOOD phase II randomized controlled trial. European Association for the Study of the Liver (EASL) Congress, 2025. Abstract LB25222
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
5 | ||||||||
•Poster #3: CLM-022, a dual inhibitor of priming and activation steps of NLRP3 inflammasome, as a potential treatment for acute and chronic inflammatory late-stage liver diseases
•Poster #4: Intraperitoneal administration of investigational drug VS-01 captures accumulated metabolites in patients with acutely decompensated liver cirrhosis
The fifth poster was related to GENFIT's latest work on Real-World data in ACLF:
•Poster #5: A Machine-Learning algorithm using Real-World Data identified subpopulations at risk for adverse outcomes in patients with Acute-on-Chronic Liver Failure
The sixth poster was related to GENFIT's diagnostic tool NIS2+® in MASH:
•Poster #6: Development and validation of two NIS2+®-based models for the detection of MASH resolution and fibrosis improvement
2.Events reflecting the growing importance of ACLF during the EASL Congress
EASL Studio9
GENFIT joined an EASL Studio session moderated by Professor Debbie Shawcross, EASL General Secretary. She was joined by EASL Studio Editorial Board Panel Representative, Thomas Reiberger, Professor of Hepatology at the Medical University of Vienna, to discuss the topic: “Moving towards recompensation: Putting out the fire that fuels decompensation and ACLF.” Industry representatives were GENFIT and Boehringer Ingelheim.
EF CLIF & GENFIT: “Continuing Our Mission to Address ACLF”
In accordance with the partnership established in 2024, GENFIT and the European Foundation for the Study of Chronic Liver Failure (EF CLIF) co-hosted a session, to explore the latest scientific insights and collaborative efforts in the presence of:
•Dr Arun J Sanyal, Professor of Medicine and Director of the Division of Gastroenterology at the Medical Center of the Virginia Commonwealth University in Richmond, USA. Former President of the American Association for the Study of Liver Diseases (AASLD);
•Professor Pierre-Emmanuel Rautou, Clinical Specialist in Hepatology, Professor of Hepatology at Université Paris-Cité and Beaujon Hospital (Clichy, France), Head of the splanchnic hemodynamic laboratory at Beaujon Hospital;
•Professor Richard Moreau, Chairman of the Scientific Advisory Board of EF CLIF and Professor of Hepatology, Liver Unit, Beaujon Hospital, Assistance Publique Hôpitaux de Paris (APHP), Clichy, France.
On this occasion, Dean Hum, Chief Scientific Officer at GENFIT, provided an update on the pipeline and R&D initiatives comprising GENFIT’s ACLF franchise.
ACLF Patient Advocacy Council
GENFIT hosted a ACLF Patient Advocacy Council session, involving:
•Dr. Jennifer Lai, leading transplant hepatologist from University of California San Francisco (MD, MBA, FACP);
•Patient advocacy groups: Global Liver Institute (GLI) and European Liver Patients’ Association (ELPA);
•Two patients who have had a liver transplant as a treatment for ACLF, and one patient caregiver.
Discussions focused on ACLF awareness, information gaps, awareness of clinical trials, and regulatory engagement, from a patient advocacy perspective.
Roundtable discussion “Advancing the Use of AI Tools in Hepatology Drug Development”
GENFIT participated in a roundtable discussion organized by Scrip-CITELINE on “Advancing the Use of AI Tools in Hepatology Drug Development” to take place on May 8, 2025.
CCA: Acquisition of the full intellectual property rights for GNS561 from Genoscience Pharma | ||||||||
In early 2025, GENFIT completed the acquisition of the full intellectual property rights for GNS561 from Genoscience Pharma, expanding upon the limited rights initially obtained through a license at the end of 2021.
With this agreement, GENFIT acquired all patents and patent applications, know-how, and data held by Genoscience Pharma necessary for the development, manufacturing, and marketing of GNS561 worldwide (“GNS561 Technology”) regardless of its therapeutic indication, form, dosage, or formulation, incorporating in whole or in part the GNS561 Technology (including as an active ingredient) or manufactured using this Technology. As a result of the acquisition, the license agreement signed in 2021 was terminated.
9 EASL Studio is the first platform created by EASL to facilitate innovative scientific and strategic discussions. It brings together a wide range of stakeholders, including academics, industry partners, policymakers, regulatory bodies, and patients, to discuss a selected topic from various perspectives.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
6 | ||||||||
As a reminder, at the end of 2021, we signed a license from Genoscience Pharma to develop and commercialize the investigational treatment GNS561 in cholangiocarcinome (CCA) in the United States, Canada and Europe, including the United Kingdom and Switzerland.
Genoscience Pharma is a French clinical-stage biotechnology company developing novel lysosomotropic therapeutics to establish a new standard of care against cancer, autoimmune and infectious diseases.
For more information about the financial terms of this new agreement, see Note 2.1 to our consolidated financial statements included in this report.
Main events related to Corporate Governance | ||||||||
Following the retirement of Chief Medical Officer (CMO) Carol Addy on June 30, 2025, a new CMO has been appointed and will officially assume the role at the end of the year.
Chief Scientific Officer Dean Hum will retire, effective as of September 30, 2025. He will be replaced by Sakina Sayah-Jeanne, currently EVP Research & Translational Science and member of the Executive Committee since she joined GENFIT in 2022.
In order to strengthen its composition and its expertise in financial and extra-financial matters, the Board of Directors proposed to shareholders the appointment of Mr. Tristan Imbert as director. The nomination was approved for a three-year term by the Shareholders' Meeting held on June 17, 2025. Mr. Tristan Imbert joined the Audit Committee and the ESG Committee following his appointment.
The biography of Mr. Tristan Imbert, including the positions he has held, is provided below:
 | Tristan Imbert Independent Director of the Company Mr Tristan Imbert is an experienced CFO with over 18 years' experience in finance, 10 years in R&D, and 7 years in strategic roles. Mr. Tristan Imbert began his career at Sanofi Aventis in the R&D function and was based in France, the UK and the USA (1989-2000). In 2000 he joined the consulting firm BCG. Based between Paris and New York, he worked with clients in the pharmaceutical industry. In 2005, Tristan Imbert joined Novartis and was appointed Head of Strategic Planning in Basel. He progressed within the Group in the finance function and became CFO Nordic Countries and then CFO Canada. In 2018, he was promoted to CFO of Novartis Gene Therapies. After three and a half years in this role, he joined biotech Cimeio Therapeutics in 2021, as CFO, and prepared the company for a potential IPO and new financing round. Mr. Tristan Imbert left Cimieo Therapeutics in 2024 and no longer has an operational role. He worked in North America (Canada and the United States) and Europe (France, Switzerland, Sweden, and the United Kingdom) in international and multicultural environments. Tristan Imbert holds a Master's degree in applied mathematics from the Université de Paris-Sud and an MBA from Columbia University-Graduate School of Business in New York. | |||||||
The terms of office of Mr. Eric Baclet and Ms. Katherine Kalin were renewed for a period of three years at the same Annual Shareholders Meeting.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
7 | ||||||||
At the date of this report, GENFIT's Board of Directors and its committees are as follows:
| Independent Director | Audit Committee | Nomination and Compensation Committee | Strategy and Alliances Committee | ESG Committee | |||||||||||||
Jean-François Mouney Chairman of the Board | No | Member | Chairman | Member | |||||||||||||
Éric Baclet Vice-Chairman of the Board | Yes | Member | Chairman | ||||||||||||||
Florence Séjourné (representative of Biotech Avenir SAS) Director | No | ||||||||||||||||
Sandra Silvestri (representative of IPSEN) Director | No | ||||||||||||||||
Katherine Kalin Director | Yes | Member | Member | ||||||||||||||
Catherine Larue Director | Yes | Member | Chairman | ||||||||||||||
Anne-Hélène Monsellato Director | Yes | Chairman | |||||||||||||||
Jean-François Tiné Director | Yes | Member | Member | ||||||||||||||
Tristan Imbert Director | Yes | Member | Member | ||||||||||||||
At the Company’s Annual Shareholders’ Meeting held on June 17, 2025, all of the resolutions recommended by the Board of Directors were adopted by a significant majority of the votes cast; this includes the renewal of financial authorizations that would allow the Company flexibility to seize relevant market opportunities.
ESG commitment | ||||||||
In 2024, GENFIT has been monitoring regulatory developments and has prepared a progressive compliance plan for non-financial reporting under the CSRD framework, until the draft of Omnibus Directive published at the beginning of 2025 led to the suspension of our transition plan to the ESRS standards associated with this directive. This regulatory development does not, however, call into question our desire to move closer to European standards, as part of a voluntary approach tailored to our corporate profile. GENFIT will therefore continue to monitor best reporting practices and adjust as needed.
In May 2025, GENFIT published its annual Extra-Financial Performance Report (fiscal year 2024), highlighting its latest initiatives and providing insights on the evolution of key performance indicators.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
8 | ||||||||
| 2.2 | Strategy and Outlook | |||||||
| Our approach to generate value | ||||||||
In drug development, our goal is to focus our efforts in one specific area - rare and life-threatening liver diseases - for greater operational efficiency, and to distribute risk across different programs with different mechanisms of action, with the goal to improve our chances of success.
Our goal is also to reduce development timelines, and we therefore favor two approaches to strengthen our portfolio:
•Repurposing of molecules approved in other indications (e.g. NTZ, an antiparasitic drug, in ACLF and revised as part of our G1090N program)
•In-licensing and/or acquisition of molecules initially developed by other companies (e.g. GNS561, for which the intellectual property rights were acquired from Genoscience Pharma; ACLF pipeline : VS-02, SRT-015 and CLM-022, respectively, through the ownership of Versantis AG, the license agreement with Seal Rock Therapeutics and the license agreement reached with Celloram Inc.).
GENFIT's ambition is to develop drug candidates from the earliest stages up to the latest stages, including Phase 3 and pre-commercialization. Depending on predefined criteria such as the targeted indication or competitive environment, or potential opportunities in terms of partnerships, GENFIT will then choose what we consider to be the best option to commercialize our most promising assets for which the Company has not yet out-licensed the rights:
•Build our own marketing and sales forces to commercialize the asset on our own, or
•Leverage the existing relationship with our preferred commercial partner Ipsen which provides a natural path to commercialization, or
•Commercialize via another partner.
We consider the patient journey as a whole and are also looking to continue to be present in the diagnostic field, specifically to determine which populations to treat within the therapeutic areas we are targeting with our drug candidates.
Our corporate priorities for 2025 | ||||||||
In accordance to this strategy, GENFIT continues, during the second half of 2025, to prioritize the execution of its clinical development programs underway at the date of this report, as well as research programs focused on pre-clinical/non-clinical development.
G1090N (new formulation of NTZ) is now GENFIT's lead asset in ACLF, and the aim is now to deliver safety data and early markers of efficacy on healthy volunteers by the end of the year.
With regard to the GNS561 program in CCA, the aim is to continue with the on-going Phase 1b clinical trial and deliver data by the end of the year.
Following the discontinuation of our VS-01 program in ACLF, we anticipate a substantial reduction in our operating expenses. This will provide strategic flexibility, and extended cash runway might be used as a means to explore new mechanistic approaches through early-stage business development initiatives aiming to tackle the multiple dimensions of the urgent gaps in ACLF care.
| Impact on financial outlook | ||||||||
Following the discontinuation of our VS-01 program in ACLF, we anticipate a substantial reduction in our operating expenses. We expect that our existing cash and cash equivalents will enable us to fund our operating expenses and capital expenditure requirements beyond the end of 2028, enabling the Company to further develop its pipeline focused on Acute-on-Chronic Liver Failure (ACLF) and support general corporate purposes. This estimation is based on current assumptions and programs and does not include exceptional events. This estimation assumes i) our expectation to receive significant future commercial milestone revenue pursuant to the license agreement with Ipsen and Ipsen meeting its sales-based thresholds, ii) drawing down all additional installments under the Royalty Financing, and iii) the reimbursement at maturity in October 2025 of any OCEANEs not converted or repurchased and cancelled10.
10 For a total of €586 thousand as of the date of this report.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
9 | ||||||||
| 2.3 | Operating and Financial Review | |||||||
| 2.3.1 | Comments on the condensed statement of net income for the periods ended June 30, 2024 and June 30, 2025 | |||||||
Revenue and other income | ||
The Company’s revenue and other income mainly comprises revenue, the research tax credit, and other operating revenue.
| Revenues and other income | Half-year ended | |||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | ||||||
| Revenues | 58,973 | 33,488 | ||||||
| CIR tax credit | 2,108 | 2,030 | ||||||
| Government grants and subsidies | 21 | 17 | ||||||
| Other operating income | 97 | 135 | ||||||
| TOTAL | 61,199 | 35,670 | ||||||
Revenues are composed of:
| Revenues | Half-year ended | |||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | ||||||
Royalty revenue | 154 | 6,871 | ||||||
Milestone revenue | 48,686 | 26,556 | ||||||
Revenue initially deferred from the Licensing Agreement (Ipsen) | 9,354 | 0 | ||||||
Revenue from the Part B Transition Services Agreement (Ipsen) | 752 | 0 | ||||||
Other revenue | 28 | 61 | ||||||
| TOTAL | 58,973 | 33,488 | ||||||
Royalty revenue
Royalty revenue is derived from sales of Ipsen's Iqirvo® (elafibranor). These are utilized to repay the Group's Royalty Financing agreement. See Note 17 - Loans and borrowings.
Milestone revenue
On May 20, 2025, GENFIT announced that Ipsen’s Iqirvo® (elafibranor) was granted pricing and reimbursement in Italy for Primary Biliary Cholangitis (PBC), the third major European country to do so in addition to the UK and Germany. This third approval triggered a new milestone payment of €26.5 million under GENFIT's Licensing and Collaboration Agreement with Ipsen, due upon pricing and reimbursement of Iqirvo® (elafibranor) in three major European markets.
Other income is composed of:
| Other income | Half-year ended | |||||||||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | ||||||||||||
| CIR tax credit | 2,108 | 2,030 | ||||||||||||
| Other operating income (including exchange gains on trade payables and receivables) | 97 | 135 | ||||||||||||
| Government grants and subsidies | 21 | 17 | ||||||||||||
| TOTAL | 2,226 | 2,182 | ||||||||||||
CIR tax credit
During the first six months of 2025, the research tax credit (CIR) amounted to €2,030 (€2,108 for the same period in 2024), due to a reduction in eligible research and development expenses.
Other operating income
During the first six months of 2025, the Group recognized €135 in “Other operating income” (€97 for the same period in 2024), mainly comprised of exchange gains on trade payables and trade receivables.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
10 | ||||||||
Operating Expenses by destination | ||
The tables below break operating expenses down by destination, mainly into research and development expenses, general and administrative expenses, and marketing and market access expenses.
| Operating expenses and other operating income (expenses) | Half-year ended | Of which : | ||||||||||||||||||||||||||||||||||||||||||
| 2024/06/30 | Raw | Contracted | Employee | Other | Depreciation, | Gain / | ||||||||||||||||||||||||||||||||||||||
| materials | research and | expenses | expenses | amortization | (loss) on | |||||||||||||||||||||||||||||||||||||||
| and | development | (maintenance, | and | disposal of | ||||||||||||||||||||||||||||||||||||||||
| consumables | activities | fees, travel, | impairment | property, | ||||||||||||||||||||||||||||||||||||||||
| used | conducted by | taxes…) | charges | plant and | ||||||||||||||||||||||||||||||||||||||||
| (in € thousands) | third parties | equipment | ||||||||||||||||||||||||||||||||||||||||||
| Research and development expenses | (18,984) | (1,056) | (7,838) | (6,610) | (2,806) | (675) | — | |||||||||||||||||||||||||||||||||||||
| General and administrative expenses | (10,564) | (152) | (69) | (4,380) | (5,778) | (185) | — | |||||||||||||||||||||||||||||||||||||
| Marketing and market access expenses | (390) | (2) | — | (295) | (89) | (3) | — | |||||||||||||||||||||||||||||||||||||
| Other operating income (expenses) | (39) | — | — | — | (102) | — | 62 | |||||||||||||||||||||||||||||||||||||
| TOTAL | (29,977) | (1,210) | (7,907) | (11,284) | (8,774) | (863) | 62 | |||||||||||||||||||||||||||||||||||||
| Operating expenses and other operating income (expenses) | Half-year ended | Of which : | ||||||||||||||||||||||||||||||||||||||||||
| 2025/06/30 | Raw | Contracted | Employee | Other | Depreciation, | Gain / | ||||||||||||||||||||||||||||||||||||||
| materials | research and | expenses | expenses | amortization | (loss) on | |||||||||||||||||||||||||||||||||||||||
| and | development | (maintenance, | and | disposal of | ||||||||||||||||||||||||||||||||||||||||
| consumables | activities | fees, travel, | impairment | property, | ||||||||||||||||||||||||||||||||||||||||
| used | conducted by | taxes…) | charges | plant and | ||||||||||||||||||||||||||||||||||||||||
| (in € thousands) | third parties | equipment | ||||||||||||||||||||||||||||||||||||||||||
| Research and development expenses | (25,117) | (1,083) | (13,377) | (7,339) | (2,592) | (726) | — | |||||||||||||||||||||||||||||||||||||
| General and administrative expenses | (9,971) | (155) | (61) | (4,410) | (5,112) | (233) | — | |||||||||||||||||||||||||||||||||||||
| Marketing and market access expenses | (392) | (4) | — | (362) | (23) | (3) | — | |||||||||||||||||||||||||||||||||||||
| Other operating income (expenses) | (115) | — | — | — | (127) | — | 12 | |||||||||||||||||||||||||||||||||||||
| TOTAL | (35,594) | (1,241) | (13,439) | (12,111) | (7,854) | (961) | 12 | |||||||||||||||||||||||||||||||||||||
Research and development expenses
For the first six months of 2024, research and development expenses totaled €19.0 million. These expenses were comprised of €7.8 million in contracted research and development conducted by third parties, €6.6 million in employee expenses, €2.8 million in other expenses, €0.7 million in depreciation, amortization and impairment charges and €1.1 million in raw materials and consumables.
For the first six months of 2025, research and development expenses totaled €25.1 million. These expenses were comprised of €13.4 million in contracted research and development conducted by third parties, €7.3 million in employee expenses, €2.6 million in other expenses, €0.7 million in depreciation, amortization and impairment charges and €1.1 million in raw materials and consumables.
The increase of €5.5 million in contracted research and development conducted by third parties is mainly due to:
•Increasing costs related to the VS-01 product candidate of €5.8 million,
•Increasing costs related to the G1090N product candidate of €0.7 million,
•Decreasing costs related to the GNS561 product candidate of €0.2 million, and
•No further costs related to the ELATIVE® product candidate (approved by the FDA in the US in June 2024 and marketed under the name Iqirvo® (elafibranor) for a total variance of €0.8 million, inclusive of accrual reversals made in 2025.
The increase of €0.7 million in employee expenses, consisting of wages, salaries, social security, pension costs and share-based compensation paid to employees in the research and development function, relates primarily to the increase in workforce (from 106 to 122 employees at June 30, 2024 and 2025, respectively).
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
11 | ||||||||
The decrease of €0.2 million in other expenses is mainly due to increasing costs related to maintenance costs of €0.3 million, decreasing costs related to consultants of €0.4 million, and decreasing costs related to shipping and logistics of €0.1 million.
General and administrative expenses
For the first six months of 2024, general and administrative expenses totaled €10.6 million. These expenses were mainly comprised of €4.4 million in employee expenses and €5.8 million in other expenses.
For the first six months of 2025, general and administrative expenses totaled €10.0 million. These expenses were mainly comprised of €4.4 million in employee expenses and €5.1 million in other expenses.
The decrease of €0.7 million in other expenses in the general and administrative function was mainly due to decreases in i) donations of €0.2 million, ii) patent maintenance expenses of €0.2 million, iii) consultants of €0.2 million, and iv) recruiting fees of €0.1 million.
Marketing and market access expenses
For the first six months of 2024, marketing and market access expenses totaled €0.4 million.
For the first six months of 2025, marketing and market access expenses totaled €0.4 million.
Marketing and market access expenses remained stable period over period.
Financial income (expenses) | ||
For the half-year ended June 30, 2025, financial income amounted to loss of €10.2 million, compared to a loss of €0.9 million for the same period in 2024.
| Financial income and expenses | Half-year ended | |||||||||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | ||||||||||||
| Financial income | ||||||||||||||
| Interest income | 535 | 295 | ||||||||||||
| Foreign exchange gain | 271 | 322 | ||||||||||||
| Financial income from OCEANE repurchase | 0 | 326 | ||||||||||||
| Other financial income | 740 | 906 | ||||||||||||
| TOTAL - Financial income | 1,546 | 1,850 | ||||||||||||
| Financial expenses | ||||||||||||||
| Interest expenses | (2,327) | (1,126) | ||||||||||||
| Interest expenses for leases | (33) | (27) | ||||||||||||
| Royalty financing debt issuance costs | 0 | (4,020) | ||||||||||||
| Changes in fair value through profit or loss | 0 | (5,434) | ||||||||||||
| Foreign exchange losses | (42) | (1,399) | ||||||||||||
| Other financial expenses | (18) | (21) | ||||||||||||
| TOTAL - Financial expenses | (2,419) | (12,027) | ||||||||||||
| FINANCIAL GAIN (LOSS) | (873) | (10,178) | ||||||||||||
For the first six months of 2024, this is primarily the result of interest expense of €2.4 million, realized and unrealized net foreign exchange gain of €0.2 million, and in accrued and realized interest income €1.3 million.
For the first six months of 2025, this is primarily the result of interest expense of €1.3 million, realized and unrealized net foreign exchange loss of €1.1 million, accrued and realized interest income €1.2 million in, a one-time gain related to the OCEANEs repurchase of €0.3 million, Royalty Financing issuance costs of €4.0 million, and changes in fair value related to the Royalty Financing liability of €5.4 million.
Net income (loss) | ||
The first half of 2025 resulted in net loss of €9,956 thousand compared with a net profit of €30,311 thousand in the first half of 2024.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
12 | ||||||||
2.3.2 | Comments on the Group’s Cash Flows for the periods ended June 30, 2024 and June 30, 2025 | |||||||
As of June 30, 2025, cash and cash equivalents amounted to €107,511 (€81,788 as of December 31, 2024).
Over the period, changes in cash flow by type of flow were as follows:
| Half-year ended | ||||||||||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | ||||||||||||
| Cash flows provided by (used in) operating activities | (11,187) | (30,102) | ||||||||||||
| Cash flows provided by (used in) investment activities | (687) | (3,219) | ||||||||||||
| Cash flows provided by (used in) financing activities | (4,225) | 59,287 | ||||||||||||
Cash flows provided by (used in) operating activities | ||
Cash flow used in operating activities amounted to an outflow of €30,102 thousand for the half-year ended June 30, 2025 compared with an outflow of €11,187 thousand for the half-year ended June 30, 2024.
In the first half of 2025, this amount mainly stems from our research and development efforts; UNVEIL-IT®, our Phase 2 clinical trial of VS-01 in ACLF (and related proof-of-concept study in said indication); GNS561, as part of its CCA program; NTZ, as part of its ACLF program; SRT-015, as part of its ACLF program; and CLM-22, as part of its ACLF program.
In the first half of 2024, this amount mainly stems from our research and development efforts; notably for ELATIVE®, our Phase 3 clinical trial of elafibranor in PBC (approved by the FDA in June 2024); UNVEIL-IT®, our Phase 2 clinical trial of VS-01 in ACLF; GNS561, as part of its Cholangiocarcinoma program; and NTZ, as part of its ACLF program.
These cash flows reflect GENFIT’s business, which requires significant research and development efforts, and generates expenses that change in line with progress on the Company’s research programs, net of its operating revenues.
Cash flows provided by (used in) investing activities | ||
Cash flow used in investing activities amounted to €(3,219) thousand in the first half of 2025, compared with €(687) thousand in cash flow provided in the first half of 2024.
In the first half of 2025, these cash flows include acquisitions and disposals of fixed assets and financial assets, including the €2 million acquisition of all patents and patent applications, know-how, and data held by Genoscience Pharma necessary for the development, manufacturing, and marketing of GNS561, regardless of its therapeutic indication, form, dosage, or formulation. For further information please refer to Note 2.1 - "Major events in the period" of section 3.6 - Notes to the consolidated financial statements.
Cash flows provided by (used in) financing activities | ||
Cash flow used in financing activities amounted to €59,287 thousand in the first half of 2025, compared with €(4,225) thousand in the first half of 2024.
In the first half of 2025, these cash flows mainly reflect the Royalty Financing agreement (receipt of €130 million) and the OCEANEs repurchase (payment of €61.7 million). For further information please refer to Note 2.1 - "Major events in the period" of section 3.6 - Notes to the consolidated financial statements.
Currencies | ||
GENFIT has expenses and owns bank accounts in multiple currencies, including the Euro (EUR), the US Dollar (USD) and the Swiss Franc (CHF). For further information please refer to Note 6.1 - "Foreign exchange risk" of section 3.6 - Notes to the consolidated financial statements.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
13 | ||||||||
| 2.4 | Main Transactions with Related Parties | |||||||
Investors are invited to refer to the information provided in Item 7.B - Related Party Transaction and Note 28 to the Consolidated Annual Financial Statements for the year ended December 31, 2024 in the 2024 Annual Report on Form 20-F (the "2024 Form 20-F") for a summary of the Company's principal ongoing transactions with related parties. Transactions with related parties occurring during the first half of 2025 are described in Note 23 - "Related parties" of the half year condensed consolidated financial states for the period ended June 30, 2025 included in section 3 of this report.
| 2.5 | Main Risks and Uncertainties | |||||||
We encourage investors to take into consideration all of the information presented in our 2024 Form 20-F and in this Half-Year Business and Financial Report before deciding to invest in Company shares. This includes, in particular, the risk factors described in Item 3.D. “Risk Factors” of the 2024 Form 20-F (and the contents of this section), of which the realization may have (or has had in some cases) material adverse effect on the Group and its activity, financial situation, results, development or perspectives, and which are of importance in the investment decision-making process.
With the exception of the following risk factors, which are updated and replaced as below, our review of our risk factors has not prompted any modifications in the nature, quantity or categories of risk factors, nor in their ranking in terms of probability of occurrence or impact, in comparison with what was presented in Item 3.D “Risk Factors” of the 2024 Form-20-F. The risks faced by the Company and described in the 2024 Form 20-F remain essentially the same.
Drug development is subject to a number of risks and the Group is highly exposed to the occurrence of any one of these inherent risks. Our activities in this area are all the more risky as many of our drug candidates are being evaluated in ACLF, a new therapeutic area characterized by a life-threatening condition that may be fatal in the short term, are at an early development stage and, for some of them, we were not involved in the initial research and discovery work, and may be less familiar with their mechanisms of action.
Drug development is a long, costly and uncertain process, aimed at demonstrating the therapeutic benefit of a drug candidate that competes with existing products and standards of care or other drug candidates in development.
Since the successful clinical development of Iqirvo® (elafibranor) (Iqirvo® is the commercial name used by our partner Ipsen to market elafibranor in PBC) carried out under the licensing agreements we signed with Terns Pharmaceuticals in 2019 in Greater China, and Ipsen in 2021 in other major pharmaceutical markets, our product pipeline is now composed of drug candidates whose development is much less advanced and therefore inherently more risky. These drug candidates, even if they have demonstrated promising initial preclinical or clinical results, have yet to obtain their preclinical and/or clinical proof-of-concept in the indications for which they are intended, and their safety and tolerability profiles are not yet proven.
For example, in the second half of 2023, our drug candidates VS-01 in ACLF and GNS561 in CCA with KRAS mutation entered Phase 2 (stopped in September 2025) and Phase 1b/2 respectively, in order to provide clinical proof-of-concept and better understand the safety and tolerability profiles of these mechanisms of action.
Our other drug candidates are at an even earlier stage, since they have either obtained initial Phase 1 clinical trial results, which is the case for NTZ before we decided to reformulate this drug candidate now referred to as G1090N, or have never been administered in humans in the therapeutic areas in which we are developing them (G1090N, SRT-015 in intravenous formulation, and CLM-022 in ACLF, VS-01 in UCD/OAs and VS-02 in HE).
For some of them — either because we are developing them as part of a repositioning strategy or because we have in-licensing or acquired the relevant intellectual property — we have developed, are in the process of developing, or will have to develop new pharmaceutical formulations that may not demonstrate the desired therapeutic efficacy and/or safety.
Most of these drug candidates are being developed to prevent and/or treat ACLF (G1090N, SRT-015 and CLM-022), a condition for which we have little experience in recruiting patients for clinical trials, for which no treatment has yet been approved and in a disease area characterized by a life-threatening condition that may be fatal in the short term. ACLF shares these characteristics with CCA with KRAS mutation. As a result, we are more exposed to the risks associated with the preclinical and clinical development of our drug candidates than companies operating in better-understood therapeutic areas, with patients suffering from less life-threatening diseases and diseases for which there are already approved treatments and clearly-defined pathways to regulatory approval. We are also exposed to the risks and uncertainties of not being able to demonstrate that our drug candidates provide sufficient therapeutic benefit. Some of these product candidates are also intended to treat diseases for which we have limited experience with drug development, which creates further risks in their development.
Finally, a significant part of our development pipeline (G1090N, VS-01 and VS-02, SRT-015 and CLM-022) results either from the acquisition of licensing rights or intellectual property from other companies (Genoscience, Seal Rock Therapeutics and Celloram), or from our Group's acquisition of Versantis AG. Despite due diligence and evaluation procedures we have carried out on the quality of previous results obtained by these companies, the development of these programs is riskier than if we had developed them ourselves from the outset.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
14 | ||||||||
3. | HALF-YEAR CONDENSED CONSOLIDATED FINANCIAL STATEMENTS AT JUNE 30, 2025 | |||||||
| 3.1 | Consolidated Statements of Financial Position | |||||||||||||
| 3.2 | Consolidated Statements of Operations | |||||||||||||
| 3.3 | Consolidated Statements of Other Comprehensive Loss | |||||||||||||
| 3.4 | Consolidated Statements of Cash Flows | |||||||||||||
| 3.5 | Consolidated Statements of Changes in Equity | |||||||||||||
| 3.6 | Notes to the Consolidated Financial Statements | |||||||||||||
| Note 1 | The Company | |||||||||||||
| Note 2 | Major Events in the Period and Events after the Period | |||||||||||||
| Note 3 | Basis of Presentation | |||||||||||||
| Note 4 | Summary of Material Accounting Information | |||||||||||||
Note 5 | Segment Information | |||||||||||||
Note 6 | Financial Risks Management | |||||||||||||
Note 7 | Revenues and Other Income | |||||||||||||
Note 8 | Operating expenses | |||||||||||||
| Note 9 | Financial Income and Expenses | |||||||||||||
| Note 10 | Income Tax | |||||||||||||
| Note 11 | Earnings (loss) Per Share | |||||||||||||
Note 12 | Cash and Cash Equivalents | |||||||||||||
Note 13 | Current Trade and Other Receivables | |||||||||||||
Note 14 | Other Current Assets | |||||||||||||
Note 15 | Intangible Assets | |||||||||||||
Note 16 | Other Non-current Financial Assets | |||||||||||||
| Note 17 | Loans and Borrowings | |||||||||||||
Note 18 | Current Trade and Other Payables | |||||||||||||
Note 19 | Current Provisions | |||||||||||||
Note 20 | Equity | |||||||||||||
Note 21 | Fair Value of Financial Instruments | |||||||||||||
Note 22 | Litigation | |||||||||||||
Note 23 | Related Parties | |||||||||||||
Note 24 | Commitments, Contingent Liabilities and Contingent Assets | |||||||||||||
Note 25 | Supplemental Cash Flow Information | |||||||||||||
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
15 | ||||||||
3.1 | Consolidated Statements of Financial Position | |||||||
| ASSETS | Notes | As of | ||||||||||||||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||||||||||||||
| Current assets | ||||||||||||||||||||
| Cash and cash equivalents | 12 | 81,788 | 107,511 | |||||||||||||||||
| Current trade and others receivables | 13 | 7,564 | 43,709 | |||||||||||||||||
| Other current assets | 14 | 3,409 | 4,204 | |||||||||||||||||
| Inventories | — | 4 | 4 | |||||||||||||||||
| Total - Current assets | 92,766 | 155,429 | ||||||||||||||||||
| Non-current assets | ||||||||||||||||||||
| Intangible assets | 15 | 47,998 | 50,346 | |||||||||||||||||
| Property, plant and equipment | 15 | 7,595 | 7,905 | |||||||||||||||||
| Other non-current financial assets | 16 | 3,065 | 3,002 | |||||||||||||||||
| Deferred tax assets | 10 | — | — | |||||||||||||||||
| Total - Non-current assets | 58,659 | 61,254 | ||||||||||||||||||
| Total - Assets | 151,424 | 216,683 | ||||||||||||||||||
| SHAREHOLDERS' EQUITY AND LIABILITIES | Notes | As of | ||||||||||||||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||||||||||||||
| Current liabilities | ||||||||||||||||||||
| Current convertible loans | 17 | 54,572 | 582 | |||||||||||||||||
| Other current loans and borrowings | 17 | 2,009 | 2,044 | |||||||||||||||||
| Current trade and other payables | 18 | 18,387 | 23,757 | |||||||||||||||||
| Current provisions | 19 | 40 | 40 | |||||||||||||||||
| Liability from royalty financing agreement | 17 | — | 15,015 | |||||||||||||||||
| Other current tax liabilities | 10 | 155 | 137 | |||||||||||||||||
| Total - Current liabilities | 75,162 | 41,575 | ||||||||||||||||||
| Non-current liabilities | ||||||||||||||||||||
| Other non-current loans and borrowings | 17 | 5,552 | 4,688 | |||||||||||||||||
| Liability from royalty financing agreement | 17 | — | 116,584 | |||||||||||||||||
| Non-current employee benefits | 1,341 | 1,364 | ||||||||||||||||||
| Deferred tax liabilities | 10 | 145 | — | |||||||||||||||||
| Total - Non-current liabilities | 7,038 | 122,636 | ||||||||||||||||||
| Shareholders' equity | ||||||||||||||||||||
| Share capital | 20 | 12,499 | 12,501 | |||||||||||||||||
| Share premium | 20 | 446,948 | 440,277 | |||||||||||||||||
| Retained earnings (accumulated deficit) | 20 | (392,077) | (390,535) | |||||||||||||||||
| Currency translation adjustment | 20 | 347 | 186 | |||||||||||||||||
| Net profit (loss) | — | 1,507 | (9,956) | |||||||||||||||||
| Total - Shareholders' equity | 69,224 | 52,472 | ||||||||||||||||||
| Total - Shareholders' equity & liabilities | 151,424 | 216,683 | ||||||||||||||||||
The accompanying notes form an integral part of these consolidated financial statements.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
16 | ||||||||
3.2 | Consolidated Statements of Operations | |||||||
| Notes | Half-year ended | |||||||||||||||||||
| (in € thousands, except earnings per share data) | 2024/06/30 | 2025/06/30 | ||||||||||||||||||
| Revenues and other income | ||||||||||||||||||||
| Revenue | 7 | 58,973 | 33,488 | |||||||||||||||||
| Other income | 7 | 2,226 | 2,182 | |||||||||||||||||
| Revenues and other income | 61,199 | 35,670 | ||||||||||||||||||
| Operating expenses and other operating income (expenses) | ||||||||||||||||||||
| Research and development expenses | 8 | (18,984) | (25,117) | |||||||||||||||||
| General and administrative expenses | 8 | (10,564) | (9,971) | |||||||||||||||||
| Marketing and market access expenses | 8 | (390) | (392) | |||||||||||||||||
| Other operating expenses | 8 | (39) | (115) | |||||||||||||||||
| Operating income (loss) | 31,222 | 76 | ||||||||||||||||||
| Financial income | 9 | 1,546 | 1,850 | |||||||||||||||||
| Financial expenses | 9 | (2,419) | (12,027) | |||||||||||||||||
| Financial profit (loss) | (873) | (10,178) | ||||||||||||||||||
| Net profit (loss) before tax | 30,349 | (10,102) | ||||||||||||||||||
| Income tax benefit (expense) | 10 | (39) | 146 | |||||||||||||||||
| Net profit (loss) | 30,311 | (9,956) | ||||||||||||||||||
| Basic and diluted earnings (loss) per share | ||||||||||||||||||||
| Basic earnings (loss) per share (€/share) | 11 | 0.61 | (0.20) | |||||||||||||||||
| Diluted earnings (loss) per share (€/share) | 11 | 0.53 | (0.20) | |||||||||||||||||
The accompanying notes form an integral part of these consolidated financial statements.
3.3 | Consolidated Statements of Other Comprehensive Income (Loss) | |||||||
| Notes | Half-year ended | |||||||||||||||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | ||||||||||||||||||
| Net profit (loss) | 30,311 | (9,956) | ||||||||||||||||||
| Actuarial gains and losses net of tax | 46 | 104 | ||||||||||||||||||
| Change in fair value of equity instruments included in financial assets and financial liabilities | 16 | (923) | (114) | |||||||||||||||||
| Other comprehensive income (loss) | ||||||||||||||||||||
| that will never be reclassified to profit or loss | (877) | (10) | ||||||||||||||||||
| Exchange differences on translation of foreign operations | 20 | (1,448) | (161) | |||||||||||||||||
| Other comprehensive income (loss) | ||||||||||||||||||||
| that are or may be reclassified to profit or loss | (1,448) | (161) | ||||||||||||||||||
| Total comprehensive income (loss) | 27,986 | (10,127) | ||||||||||||||||||
The accompanying notes form an integral part of these consolidated financial statements.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
17 | ||||||||
3.4 | Consolidated Statements of Cash Flows | |||||||
| Notes | Half-year ended | Half-year ended | ||||||||||||||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | ||||||||||||||||||
| Cash flows from operating activities | ||||||||||||||||||||
| + Net profit (loss) | 30,311 | (9,956) | ||||||||||||||||||
| Reconciliation of net loss to net cash used in operating activities | ||||||||||||||||||||
| Adjustments for: | ||||||||||||||||||||
| + Depreciation and amortization on tangible and intangible assets | 15 | 854 | 897 | |||||||||||||||||
| + Impairment and provisions | 105 | 193 | ||||||||||||||||||
| + Expenses related to share-based compensation | 334 | 242 | ||||||||||||||||||
| - Loss (gain) on disposal of property, plant and equipment | (62) | (12) | ||||||||||||||||||
| + Net finance expenses (revenue) | 9 | 542 | 6,324 | |||||||||||||||||
| + Income tax expense (benefit) | 10 | 39 | (146) | |||||||||||||||||
| + Other non-cash items | 1,687 | 590 | ||||||||||||||||||
| Operating cash flows before change in working capital | 33,809 | (1,868) | ||||||||||||||||||
| Decrease (increase) in trade receivables and other assets | 13 | (39,413) | (37,840) | |||||||||||||||||
| (Decrease) increase in trade payables and other liabilities | 18 | (5,572) | 9,606 | |||||||||||||||||
| Change in working capital | (44,984) | (28,234) | ||||||||||||||||||
| Income tax paid | (12) | — | ||||||||||||||||||
| Net cash flows provided by (used in) in operating activities | (11,187) | (30,102) | ||||||||||||||||||
| Cash flows from investment activities | ||||||||||||||||||||
| - Acquisition of other intangible assets | 15 | — | (2,034) | |||||||||||||||||
| - Acquisition of property, plant and equipment | 25 | (737) | (1,054) | |||||||||||||||||
| + Proceeds from disposal of / reimbursement of property, plant and equipment | 78 | 39 | ||||||||||||||||||
| - Acquisition of financial instruments | (28) | (170) | ||||||||||||||||||
| Net cash flows provided by (used in ) investment activities | (687) | (3,219) | ||||||||||||||||||
| Cash flows from financing activities | ||||||||||||||||||||
| + Proceeds from issue of share capital (net) | — | 17 | ||||||||||||||||||
| + Proceeds from new loans and borrowings | 17 | — | 130,020 | |||||||||||||||||
| - Repayments of loans and borrowings | 17 | (3,143) | (62,105) | |||||||||||||||||
| - Repayments of royalty financing liability | 17 | — | (4,492) | |||||||||||||||||
| - Payments of debt issuance costs | 17 | — | (3,363) | |||||||||||||||||
| - Payments on lease debts | (545) | (555) | ||||||||||||||||||
| - Financial interests paid (including finance lease) | (1,073) | (530) | ||||||||||||||||||
| + Financial interests received | 535 | 295 | ||||||||||||||||||
| Net cash flows provided by (used in ) financing activities | (4,225) | 59,287 | ||||||||||||||||||
| Increase (decrease) in cash and cash equivalents | (16,100) | 25,966 | ||||||||||||||||||
| Cash and cash equivalents at the beginning of the period | 12 | 77,789 | 81,788 | |||||||||||||||||
| Effects of exchange rate changes on cash | (43) | (243) | ||||||||||||||||||
| Cash and cash equivalents at the end of the period | 61,645 | 107,511 | ||||||||||||||||||
The accompanying notes form an integral part of these consolidated financial statements.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
18 | ||||||||
3.5 | Consolidated Statements of Changes in Equity | |||||||
(Amounts in thousands of euros, except for number of shares)
| Share capital | Share | Treasury | Retained | Currency | Net | Total | ||||||||||||||||||||||||||||||||||||||||||||
| Number | Share | premium | shares | earnings | translation | profit | shareholders' | |||||||||||||||||||||||||||||||||||||||||||
| of shares | capital | (accumulated | adjustment | (loss) | equity | |||||||||||||||||||||||||||||||||||||||||||||
| (in € thousands) | deficit) | |||||||||||||||||||||||||||||||||||||||||||||||||
| As of January 01, 2024 | 49,834,983 | 12,459 | 445,261 | (970) | (360,901) | 996 | (28,894) | 67,951 | ||||||||||||||||||||||||||||||||||||||||||
| Net profit (loss) | 30,311 | 30,311 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss) | (877) | (1,448) | (2,325) | |||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive income (loss) | — | — | — | — | (877) | (1,448) | 30,311 | 27,986 | ||||||||||||||||||||||||||||||||||||||||||
| Allocation of prior period profit (loss) | (28,894) | 28,894 | — | |||||||||||||||||||||||||||||||||||||||||||||||
| Capital increase | 71,500 | 18 | 662 | (7) | 673 | |||||||||||||||||||||||||||||||||||||||||||||
| Equity component of OCEANE net of deferred taxes | 232 | 232 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | 334 | 334 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Treasury shares | 171 | 171 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Other movements | 16 | 16 | ||||||||||||||||||||||||||||||||||||||||||||||||
| As of June 30, 2024 | 49,906,483 | 12,477 | 446,490 | (799) | (390,663) | (452) | 30,311 | 97,363 | ||||||||||||||||||||||||||||||||||||||||||
| Net profit (loss) | (28,804) | (28,804) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss) | (253) | 799 | 546 | |||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive income (loss) | — | — | — | — | (253) | 799 | (28,804) | (28,258) | ||||||||||||||||||||||||||||||||||||||||||
| Allocation of prior period profit (loss) | — | — | — | |||||||||||||||||||||||||||||||||||||||||||||||
| Capital increase | 89,702 | 22 | 405 | — | 428 | |||||||||||||||||||||||||||||||||||||||||||||
| Equity component of OCEANE net of deferred taxes | (222) | (222) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | 276 | 276 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Treasury shares | (348) | (348) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Other movements | (15) | (15) | ||||||||||||||||||||||||||||||||||||||||||||||||
| As of December 31, 2024 | 49,996,185 | 12,499 | 446,948 | (1,147) | (390,930) | 347 | 1,507 | 69,224 | ||||||||||||||||||||||||||||||||||||||||||
| Net profit (loss) | (9,956) | (9,956) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Other comprehensive income (loss) | (10) | (161) | (170) | |||||||||||||||||||||||||||||||||||||||||||||||
| Total comprehensive income (loss) | — | — | — | — | (10) | (161) | (9,956) | (10,127) | ||||||||||||||||||||||||||||||||||||||||||
| Allocation of prior period profit (loss) | 1,507 | (1,507) | — | |||||||||||||||||||||||||||||||||||||||||||||||
| Capital increase | 6,375 | 2 | 15 | — | 17 | |||||||||||||||||||||||||||||||||||||||||||||
| Equity component of OCEANE net of deferred taxes | (6,929) | (6,929) | ||||||||||||||||||||||||||||||||||||||||||||||||
| Share-based compensation | 242 | 242 | ||||||||||||||||||||||||||||||||||||||||||||||||
| Treasury shares | 44 | 44 | ||||||||||||||||||||||||||||||||||||||||||||||||
| As of June 30, 2025 | 50,002,560 | 12,501 | 440,277 | (1,102) | (389,433) | 186 | (9,956) | 52,472 | ||||||||||||||||||||||||||||||||||||||||||
The accompanying notes form an integral part of these consolidated financial statements.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
19 | ||||||||
| 3.6 | Notes to the Consolidated Financial Statements | |||||||
1.THE COMPANY
Founded in 1999 under the laws of France, GENFIT S.A. (the "Company") is a late-stage biopharmaceutical company dedicated to the discovery and development of innovative drugs and diagnostic tools in therapeutic areas of high unmet need due in particular to a lack of effective treatments or diagnostic solutions and/or an increase in patients worldwide.
The Company focuses its research and development (R&D) efforts on the potential marketing of therapeutic and diagnostic solutions to combat certain metabolic, inflammatory, autoimmune and fibrotic diseases affecting in particular the liver (such as Primary Biliary Cholangitis or PBC) and more generally gastroenterological diseases. The head office address is: 885 Avenue Eugène Avinée – 59120 Loos, France.
The consolidated financial statements of the Company include the financial statements of GENFIT S.A.; those of its wholly-owned subsidiaries, GENFIT CORP. (U.S. subsidiary) and Versantis AG (Swiss subsidiary); and a French-law trust (for the benefit of the holders of the royalty financing bonds), together referred to in these notes to the consolidated financial statements as "GENFIT" or the "Group" or “we“ or “us”. There are no non-controlling interests for any period presented herein.
2.MAJOR EVENTS IN THE PERIOD AND EVENTS AFTER THE PERIOD
2.1.Major events in the period
Approval of pricing and reimbursement of Ipsen’s Iqirvo® (elafibranor) in Italy
On May 20, 2025, GENFIT announced that Ipsen’s Iqirvo® (elafibranor) was granted pricing and reimbursement in Italy for Primary Biliary Cholangitis (PBC), the third major European country to do so in addition to the UK and Germany. This third approval triggers a new milestone payment of €26.5 million under GENFIT's Licensing and Collaboration Agreement with Ipsen, due upon pricing and reimbursement of Iqirvo® (elafibranor) in three major European markets.
Milestone payments under the Licensing and Collaboration Agreement with Ipsen are not included in the scope of our royalty financing agreement.
Non-Dilutive Royalty Financing Agreement and concurrent OCEANEs repurchase
On January 30, 2025, GENFIT announced the signing of a Royalty Financing deal with HealthCare Royalty (HCRx) providing up to €185 million non-dilutive capital, including €130 million upfront, with eligibility to receive up to €55 million in two additional installments of €30 million and €20 million, respectively, based on near-term milestones.
The terms and conditions of the OCEANEs contained a negative pledge clause which limited the ability of the Company to grant security interests to its creditors upon its present or future assets or revenues. The closing of the Royalty Financing with HCRx, which was signed and announced by GENFIT on January 30, 2025, was subject to approval of OCEANEs bondholders of an amendment to this negative pledge clause, allowing for the grant of the security interest contemplated in the Royalty Financing documentation, and other customary closing conditions.
In order to obtain approval of the royalty financing by the OCEANEs holders, GENFIT convened a general meeting of the holders on March 10, 2025. All resolutions proposed by the Company to the bondholders were approved unanimously.
As announced on February 10, 2025 and February 14, 2025, the Company proposed to all of the OCEANEs holders to enter into a Put Option Agreement, pursuant to which the Company unconditionally and irrevocably undertook to repurchase the OCEANEs of such holder at a price of EUR 32.75 per bond, subject to approval by the general meeting of the OCEANEs holders of the amendment of the terms and conditions of the OCEANEs and the closing of the Royalty Financing (the “Repurchase”). Holders had until March 19, 2025 to exercise this option.
The settlement of the Repurchase occurred on March 26, 2025. 1,882,891 OCEANEs were repurchased for a total amount paid of €61.7 million. The repurchased OCEANEs were then canceled by the Company.
For further information, refer to Note 17 - Loans and Borrowings.
Genoscience Pharma asset transfer
On January 3, 2025, GENFIT acquired all patents and patent applications, know-how, and data held by Genoscience Pharma necessary for the development, manufacturing, and marketing of GNS561, regardless of its therapeutic indication, form, dosage, or formulation. The sale price was a lump sum payment of €2 million euros excluding taxes. This agreement terminates the previous agreement signed in 2021.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
20 | ||||||||
For further information, refer to Note 15 - Intangible assets and Note 24 - Commitments, contingent liabilities and contingent assets.
2.2.Events after the period
Milestone received
GENFIT collected the €26.5 million milestone payment from Ipsen in July of 2025 (recognized in May 2025 upon the third pricing and reimbursement approval of Iqirvo® (elafibranor) in Italy, the third major European country to do so).
Adverse event related to the Versantis VS-01 intangible occurred in September 2025
On September 19, 2025, GENFIT announced its decision to discontinue its VS-01 program in ACLF (Acute-on-Chronic Liver Failure), and reprioritize its development on UCD (Urea Cycle Disorder).
GENFIT’s decision follows the occurrence in September of a peritonitis case reported as Serious Adverse Event (SAE) in the UNVEIL-IT® clinical trial evaluating VS-01 in patients with ACLF grade 1, 2 or 3a and ascites and subsequent review and feedback from the independent Data Monitoring Committee (iDMC). The committee concluded that the trial could continue but required additional data and monitoring. Despite the possibility to move ahead with the study, GENFIT decided – after considering the target population’s clinical profile as well as the implications of this type of safety signals for the benefit/risk ratio of VS-01 in this indication – to discontinue both UNVEIL-IT® and the proof-of-concept study evaluating VS-01 in patients with Acute Decompensation (AD) or ACLF grade 1 with Hepatic Encephalopathy (HE) grades 1 to 4, and ascites.
The carrying amount of the Versantis VS-01 intangible is €45.8 million in "Intangible assets" on the Statement of Financial Position as of June 30, 2025, and GENFIT will fully impair the asset in the second half of 2025. In accordance with IAS 10, this is considered on a non-adjusting event as this is a situation arising entirely after the reporting period. GENFIT is currently reviewing the other possible consequences of the decision to discontinue its VS-01 program in ACLF, the impact of which cannot yet be assessed. The corresponding impact, if any, will be recognized in the second half of 2025.
3.BASIS OF PRESENTATION
The half year consolidated financial statements of GENFIT have been prepared in accordance with International Financial Reporting Standards (“IFRS”) as issued by the International Accounting Standards Board (“IASB”) and as adopted by the European Union at June 30, 2025. Comparative information is presented for the year ended December 31, 2024 and for the half year ended June 30, 2024.
The term IFRS includes International Financial Reporting Standards ("IFRS"), International Accounting Standards (the "IAS"), as well as the Interpretations issued by the Standards Interpretation Committee (the "SIC"), and the International Financial Reporting Interpretations Committee ("IFRIC").
These consolidated half year financial statements have been prepared using the historical cost measurement basis (except for certain assets and liabilities that are measured at fair value in accordance with the IFRS general principles of fair presentation), going concern, accrual basis of accounting, consistency of presentation, materiality and aggregation.
These consolidated half year financial statements for the period ended June 30, 2025 were prepared under the responsibility of the Board of Directors that approved such statements on September 19, 2025.
The principal accounting methods used to prepare the Consolidated Financial Statements are described below.
All financial information (unless indicated otherwise) is presented in thousands of euros (€).
In accordance with European Commission Regulation 1606/2002, these consolidated interim financial statements for the six-month period ended June 30, 2025 have been prepared in accordance with IAS 34 – Interim Financial Reporting, and should be read in conjunction with the Group’s most recent annual consolidated financial statements for the year ended December 31, 2024. They do not include all the information required for a complete set of financial statements in accordance with IFRS, but a selection of notes explaining significant events and transactions with a view to understanding the changes in the Group’s financial position and performance since the most recent annual consolidated financial statements.
3.1.Changes in accounting policies and new standards or amendments
The accounting policies applicable for these consolidated half-year financial statements are the same as those applied to the most recent consolidated annual financial statements.
The following new standards are applicable from January 1, 2025, but do not have any material effect on the Group’s financial statements for the period ended June 30, 2025.
•Amendments to IAS 21 - Lack of Exchangeability
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
21 | ||||||||
3.2.Standards, interpretations and amendments issued but not yet effective
The amendments and modifications to the standards below are applicable for financial years beginning after January 1, 2026, as specified below. GENFIT is in the process of assessing whether the adoption of these amendments and modifications to the standards will have a material impact on the financial statements.
•Amendments IFRS 9 and IFRS 7 regarding the classification and measurement of financial instruments, effective in 2026 (subject to endorsement by the European Union),
•Amendments IFRS 9 and IFRS 7 regarding contracts referencing nature-dependent electricity, effective in 2026 (subject to endorsement by the European Union),
•Annual Improvements to IFRS Accounting Standards — Volume 11, effective in 2026 (subject to endorsement by the European Union), and
•IFRS 19 Subsidiaries without Public Accountability: Disclosures, effective in 2027 (subject to endorsement by the European Union).
This standard in its current state is likely to have a significant impact on the financial statements:
•IFRS 18 Presentation and Disclosures in Financial Statements, effective in 2027
However, this standard has not yet been adopted by the European Union and at this stage a comprehensive evaluation is still underway.
The Group has not identified any other standard or amendment that could have a significant impact on the consolidated financial statements.
4.SUMMARY OF MATERIAL ACCOUNTING INFORMATION
4.1.Use of estimates and judgments
In preparing these consolidated financial statements, management makes judgments, estimates and assumptions that affect the application of accounting policies and the reported amounts of assets and liabilities, incomes and expenses. Actual amounts may differ from these estimates.
The estimates and underlying assumptions are reviewed on an ongoing basis. Revisions to accounting estimates are recognized in the period in which the estimates are revised and in any future periods affected.
The estimates and underlying assumptions mainly relate to the following:
•Allocation of revenue to performance obligations per the agreement with Ipsen, Note 7 - Revenues and other income
•Research tax credits, Note 7 - Revenues and other income
•Average tax rate for the annual period, Note 10 - Income tax
•Valuation of the GNS561 assets, Note 15 - Intangible assets
•Convertible loans, Note 17 - Loans and borrowings
•Valuation of the Royalty financing, Note 17 - Loans and borrowings
4.2.Consolidation
Going concern
The consolidated financial statements were prepared on a going concern basis. The Group believes it has sufficient resources to continue operating for at least twelve months following the consolidated financial statements’ publication.
When assessing going concern, the Group’s Board of Directors considers the liquidity available at the statement of financial position date, milestones whose collection is considered highly probable, the cash spend projections for the next 12-month period as from the date of the financial statements are issued, and the availability of other funding.
Consolidated entities
The Group controls an entity when it is exposed to variable returns from its involvement with the entity, and it has the ability to affect those returns through its power over the entity.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
22 | ||||||||
The Group controls all the entities included in the scope of consolidation.
Accounting policies
The accounting policies used for these interim consolidated financial statements are the same as those used for the most recent consolidated annual financial statements.
4.3.Foreign currency
Presentation currency
The Group's presentation currency is the euro.
Foreign currency transactions
Transactions in foreign currencies are translated into the respective functional currencies of the entities of the Group at the exchange rates applicable at the transaction dates. Monetary assets and liabilities denominated in foreign currencies are translated into the functional currency at the reporting date.
The resulting exchange gains or losses are recognized in the statements of operations.
Translation of foreign subsidiary financial statements
The assets and liabilities of foreign operations having a functional currency different from the euro are translated into euros at the closing exchange rate. The income and expenses of foreign operations are translated into euros at the exchange rates effective at the transaction dates or using the average exchange rate for the reporting period unless this method cannot be applied due to significant exchange rate fluctuations.
Gains and losses arising from foreign operations are recognized in the statement of other comprehensive loss. When a foreign operation is partly or fully divested, the associated share of gains and losses recognized in the currency translation reserve is transferred to the statements of operations.
Functional currencies and exchanges rates
The functional currency of GENFIT S.A. is the euro.
The functional currency of GENFIT CORP is the U.S. dollar. The applicable exchange rates used to translate the financial statements of this entity for each of the periods are as follows:
| Half-year ended | |||||||||||||||||
| Ratio : 1 US dollars (USD) = x euros (EUR) | 2024/06/30 | 2025/06/30 | |||||||||||||||
| Exchange rate at period end | 0.93414 | 0.85324 | |||||||||||||||
| Average exchange rate for the period | 0.92490 | 0.91649 | |||||||||||||||
The functional currency of Versantis AG is the Swiss Franc. The applicable exchange rates used to translate the financial statements of this entity for each of the periods are as follows:
| Half-year ended | |||||||||||
| Ratio : 1 CH franc (CHF) = x euros (EUR) | 2024/06/30 | 2025/06/30 | |||||||||
| Exchange rate at period end | 1.03799 | 1.06986 | |||||||||
| Average exchange rate for the period | 1.04035 | 1.06237 | |||||||||
5.SEGMENT INFORMATION
Segments
The Board of Directors and Chief Executive Officer are the chief operating decision makers.
The Board of Directors and the Chief Executive Officer oversee the operations and manage the business as one segment with a single activity; namely, the research and development of innovative medicines and diagnostic solutions, the marketing of which depends on the success of the clinical development phase.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
23 | ||||||||
Revenue by geographical area | Half-year ended | ||||||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | |||||||||
| Revenue from France | 100 | % | 100 | % | |||||||
| Revenue from other countries | — | % | — | % | |||||||
| TOTAL | 100 | % | 100 | % | |||||||
For the six month period ended June 30, 2024 and 2025, substantially all revenue was generated from Ipsen, including royalties recognized in France based on Ipsen's worldwide (excluding Greater China) sales of Iqirvo® (elafibranor).
Non-current assets by geographical area | As of December 31, 2024 | As of June 30, 2025 | |||||||||||||||||||||
| (thousands of euros) | France | Switzerland | Total | France | Switzerland | Total | |||||||||||||||||
| TOTAL | 12,580 | 46,079 | 58,659 | 14,880 | 46,374 | 61,254 | |||||||||||||||||
6.FINANCIAL RISKS MANAGEMENT
The Group may be exposed to the following risks arising from financial instruments: foreign exchange risk, interest rate risk, liquidity risk and credit risk.
6.1.Foreign exchange risk
The Group's overall exposure to the foreign exchange risk depends, in particular, on:
•the currencies in which it receives its revenues;
•the currencies chosen when agreements are entered into, such as licensing agreements, or co-marketing or co-development agreements;
•the location of clinical trials on drug or biomarker candidates;
•the ability for its co-contracting parties to indirectly transfer foreign exchange risk to the Company;
•the Group’s foreign exchange risk policy; and
•the fluctuation of foreign currencies against the euro.
The Group does not use any specific hedging arrangements at this time.
The Group holds cash and cash equivalents and other financial assets primarily in euros, U.S. dollars and Swiss francs as summarized here:
| Cash, cash equivalents and financial assets | As of | |||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||
| At origin, denominated in EUR | ||||||||
| Cash and cash equivalents | 70,707 | 91,676 | ||||||
| Current and non current financial assets | 3,035 | 2,974 | ||||||
| Total | 73,742 | 94,650 | ||||||
| At origin, denominated in USD | ||||||||
| Cash and cash equivalents | 10,395 | 13,840 | ||||||
| Current and non current financial assets | 16 | 14 | ||||||
| Total | 10,411 | 13,854 | ||||||
| At origin, denominated in CHF | ||||||||
| Cash and cash equivalents | 652 | 1,892 | ||||||
| Current and non current financial assets | 14 | 14 | ||||||
| Total | 666 | 1,906 | ||||||
| At origin, denominated in CAD | ||||||||
| Cash and cash equivalents | 34 | 103 | ||||||
| Current and non current financial assets | — | — | ||||||
| Total | 34 | 103 | ||||||
| Total, in EUR | ||||||||
| Cash and cash equivalents | 81,788 | 107,511 | ||||||
| Current and non current financial assets | 3,065 | 3,002 | ||||||
| Total | 84,853 | 110,514 | ||||||
The tables below show sensitivity analyses of the Group's cash and cash equivalents and expenses in both U.S. dollars Swiss francs.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
24 | ||||||||
Sensitivity analysis of the Group's cash and cash equivalents held in US dollars | ||||||||||||||
Variation of +/- 10% of the US dollar vs the euro | As of | |||||||||||||
| (in € thousands or in US dollar thousands, as applicable) | 2024/12/31 | 2025/06/30 | ||||||||||||
| Cash and cash equivalents denominated in US dollars | 10,800 | 16,219 | ||||||||||||
| Equivalent in euros, on the basis of the exchange rate described below | 10,395 | 13,839 | ||||||||||||
| Equivalent in euros, in the event of an increase of 10% of US dollar vs euro | 11,550 | 15,377 | ||||||||||||
| Equivalent in euros, in the event of a decrease of 10% of US dollar vs euro | 9,450 | 12,581 | ||||||||||||
Sensitivity analysis of the Group's expenses transacted in US dollars | ||||||||||||||
| Variation of +/- 10% of the US dollar vs the euro | Half-year ended | |||||||||||||
| (in € thousands or in US dollar thousands, as applicable) | 2024/06/30 | 2025/06/30 | ||||||||||||
| Expenses denominated in US dollars | 6,991 | 4,187 | ||||||||||||
| Equivalent in euros, on the basis of the exchange rate described below | 6,531 | 3,572 | ||||||||||||
| Equivalent in euros, in the event of an increase of 10% of US dollar vs euro | 7,256 | 3,969 | ||||||||||||
| Equivalent in euros, in the event of a decrease of 10% of US dollar vs euro | 5,937 | 3,248 | ||||||||||||
| 2025/06/30 : Equivalent in euros, on the basis of 1 dollar US = 0,85324 euros | ||||||||||||||
| 2024/06/30 : Equivalent in euros, on the basis of 1 dollar US = 0,93414 euros | ||||||||||||||
Sensitivity analysis of the Group's cash and cash equivalents held in CH franc | |||||||||||
Variation of +/- 10% of the CH franc vs the euro | As of | ||||||||||
| (in € thousands or in CH franc thousands, as applicable) | 2024/12/31 | 2025/06/30 | |||||||||
| Cash and cash equivalents denominated in CH franc | 614 | 1,768 | |||||||||
| Equivalent in euros, on the basis of the exchange rate described below | 652 | 1,891 | |||||||||
| Equivalent in euros, in the event of an increase of 10% of CH franc vs euro | 724 | 2,101 | |||||||||
| Equivalent in euros, in the event of a decrease of 10% of CH franc vs euro | 593 | 1,719 | |||||||||
Sensitivity analysis of the Group's expenses transacted in CH franc | Half-year ended | ||||||||||
Variation of +/- 10% of the CH franc vs the euro | |||||||||||
| (in € thousands or in CH franc thousands, as applicable) | 2024/06/30 | 2025/06/30 | |||||||||
| Expenses denominated in CH franc | 1,064 | 1,613 | |||||||||
| Equivalent in euros, on the basis of the exchange rate described below | 1,104 | 1,725 | |||||||||
| Equivalent in euros, in the event of an increase of 10% of CH franc vs euro | 1,227 | 1,917 | |||||||||
| Equivalent in euros, in the event of a decrease of 10% of CH franc vs euro | 1,004 | 1,568 | |||||||||
| 2025/06/30 : Equivalent in euros, on the basis of 1 CH franc = 1,06986 euros | |||||||||||
| 2024/06/30 : Equivalent in euros, on the basis of 1 CH franc = 1,03799 euros | |||||||||||
6.2.Interest rate risk
As of June 30, 2025, the Group had bank loans only at fixed interest rates. The Group's exposure to interest rate risk through its financial assets is also insignificant since these assets are mainly euro-denominated Undertakings for the Collective Investment of Transferable Securities (UCITs), medium-term negotiable notes, or term deposits with progressive rates denominated in euros or US dollars.
The Royalty Financing Bonds issued by GENFIT do not bear interest.
6.3.Liquidity risk
As of December 31, 2024 and June 30, 2025, the Group's financial liabilities totaled €62.1 million and €138.9 million.
The Group's financial liabilities at June 30, 2025 mainly consist of a Royalty Financing agreement. The reimbursement of these bonds will be tied to a portion of the royalties GENFIT receives under the Ipsen agreement from October 1, 2024. There are two specific liquidity risks related to the Royalty Financing agreement:
1.If GENFIT's royalty revenue based on sales of Iqirvo® (elafibranor) were not to achieve certain thresholds, this would hinder GENFIT's ability to draw down the remaining €55 million in two additional installments of €30 million and €20 million, respectively.
2.If GENFIT's royalty revenue based on sales of Iqirvo® (elafibranor) were to be insufficient to fully repay the Royalty Financing agreement based on the contractual multiple of 155% by December 31, 2030, this would trigger an increase in the contractual multiple (i.e., 195% or 250%) based on new reimbursement deadlines.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
25 | ||||||||
The Company has conducted a specific review of its liquidity risk and considers that it is able to meet its future maturities. On December 31, 2024 and June 30, 2025, the Group had €81,788 and €107,511 respectively in cash and cash equivalents. The Company does not believe it is exposed to short-term liquidity risk. The Company believes that the Group's cash and cash equivalents and current financial instruments are sufficient to ensure its financing for the next 12 months, in light of its current projects and obligations.
If the Group's funds were insufficient to cover any additional financing needs, the Group would require additional financing. The conditions and arrangements for any such new financing would depend, among other factors, on economic and market conditions that are beyond the Group's control.
6.4Credit risk
Credit risk is the risk of financial loss if a customer or counterparty to a financial asset defaults on their contractual commitments. The Group is exposed to credit risk due to trade receivables and other financial assets.
The Group's policy is to manage this risk by transacting with third parties with good credit standards.
7.REVENUES AND OTHER INCOME
Financial statement line item detail
| Revenues and other income | Half-year ended | |||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | ||||||
| Revenues | 58,973 | 33,488 | ||||||
| CIR tax credit | 2,108 | 2,030 | ||||||
| Government grants and subsidies | 21 | 17 | ||||||
| Other operating income | 97 | 135 | ||||||
| TOTAL | 61,199 | 35,670 | ||||||
7.1.Revenues from contracts with customers
Revenues are composed of:
Revenues | Half-year ended | |||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | ||||||
Royalty revenue | 154 | 6,871 | ||||||
Milestone revenue | 48,686 | 26,556 | ||||||
Revenue initially deferred from the Licensing Agreement (Ipsen) | 9,354 | 0 | ||||||
Revenue from the Part B Transition Services Agreement (Ipsen) | 752 | 0 | ||||||
Other revenue | 28 | 61 | ||||||
| TOTAL | 58,973 | 33,488 | ||||||
Royalty revenue
Royalty revenue is derived from worldwide sales (excluding Greater China) of Ipsen's Iqirvo® (elafibranor). These are utilized to repay the Group's Royalty Financing agreement. See Note 17 - Loans and borrowings.
Milestone revenue
On May 20, 2025, GENFIT announced that Ipsen’s Iqirvo® (elafibranor) was granted pricing and reimbursement in Italy for Primary Biliary Cholangitis (PBC), the third major European country to do so in addition to the UK and Germany. This third approval triggered a new milestone payment of €26.5 million under GENFIT's Licensing and Collaboration Agreement with Ipsen, due upon pricing and reimbursement of Iqirvo® (elafibranor) in three major European markets.
7.2.Other income
Other income is composed of:
| Other income | Half-year ended | |||||||||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | ||||||||||||
| CIR tax credit | 2,108 | 2,030 | ||||||||||||
| Other operating income (including exchange gains on trade payables and receivables) | 97 | 135 | ||||||||||||
| Government grants and subsidies | 21 | 17 | ||||||||||||
| TOTAL | 2,226 | 2,182 | ||||||||||||
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
26 | ||||||||
CIR tax credit
The Research Tax Credit ("Crédit d'Impôt Recherche," or "CIR") is granted to entities by the French tax authorities in order to encourage them to conduct technical and scientific research. Entities that demonstrate that their research expenditures meet the required CIR criteria receive a tax credit that may be used for the payment of their income tax due for the fiscal year in which the expenditures were incurred, as well as in the next three years. If taxes due are not sufficient to cover the full amount of tax credit at the end of the three-year period, the difference is paid in cash to the entity by the tax authorities. If a company meets certain criteria in terms of sales, headcount or assets to be considered a small/mid-size company, immediate payment of the Research Tax Credit can be requested. The Group meets such criteria.
The Group applies for CIR for research expenditures incurred in each fiscal year and recognizes the amount claimed in the line item "Other income" in the statements of operations in the same fiscal year. In the notes to the financial statements, the amount claimed is recognized under the heading "Research tax credit" (see Note 13 - Current trade and other receivables and the table above).
During the first six months of 2025, the research tax credit (CIR) amounted to €2,030 in 2025, (€2,108 for the same period in 2024), due to a reduction in eligible research and development expenses.
8.OPERATING EXPENSES
Financial statement line item detail
| Operating expenses and other operating income (expenses) | Half-year ended | Of which : | ||||||||||||||||||||||||||||||||||||||||||
| 2024/06/30 | Raw | Contracted | Employee | Other | Depreciation, | Gain / | ||||||||||||||||||||||||||||||||||||||
| materials | research and | expenses | expenses | amortization | (loss) on | |||||||||||||||||||||||||||||||||||||||
| and | development | (maintenance, | and | disposal of | ||||||||||||||||||||||||||||||||||||||||
| consumables | activities | fees, travel, | impairment | property, | ||||||||||||||||||||||||||||||||||||||||
| used | conducted by | taxes…) | charges | plant and | ||||||||||||||||||||||||||||||||||||||||
| (in € thousands) | third parties | equipment | ||||||||||||||||||||||||||||||||||||||||||
| Research and development expenses | (18,984) | (1,056) | (7,838) | (6,610) | (2,806) | (675) | — | |||||||||||||||||||||||||||||||||||||
| General and administrative expenses | (10,564) | (152) | (69) | (4,380) | (5,778) | (185) | — | |||||||||||||||||||||||||||||||||||||
| Marketing and market access expenses | (390) | (2) | — | (295) | (89) | (3) | — | |||||||||||||||||||||||||||||||||||||
| Other operating income (expenses) | (39) | — | — | — | (102) | — | 62 | |||||||||||||||||||||||||||||||||||||
| TOTAL | (29,977) | (1,210) | (7,907) | (11,284) | (8,774) | (863) | 62 | |||||||||||||||||||||||||||||||||||||
| Operating expenses and other operating income (expenses) | Half-year ended | Of which : | ||||||||||||||||||||||||||||||||||||||||||
| 2025/06/30 | Raw | Contracted | Employee | Other | Depreciation, | Gain / | ||||||||||||||||||||||||||||||||||||||
| materials | research and | expenses | expenses | amortization | (loss) on | |||||||||||||||||||||||||||||||||||||||
| and | development | (maintenance, | and | disposal of | ||||||||||||||||||||||||||||||||||||||||
| consumables | activities | fees, travel, | impairment | property, | ||||||||||||||||||||||||||||||||||||||||
| used | conducted by | taxes…) | charges | plant and | ||||||||||||||||||||||||||||||||||||||||
| (in € thousands) | third parties | equipment | ||||||||||||||||||||||||||||||||||||||||||
| Research and development expenses | (25,117) | (1,083) | (13,377) | (7,339) | (2,592) | (726) | — | |||||||||||||||||||||||||||||||||||||
| General and administrative expenses | (9,971) | (155) | (61) | (4,410) | (5,112) | (233) | — | |||||||||||||||||||||||||||||||||||||
| Marketing and market access expenses | (392) | (4) | — | (362) | (23) | (3) | — | |||||||||||||||||||||||||||||||||||||
| Other operating income (expenses) | (115) | — | — | — | (127) | — | 12 | |||||||||||||||||||||||||||||||||||||
| TOTAL | (35,594) | (1,241) | (13,439) | (12,111) | (7,854) | (961) | 12 | |||||||||||||||||||||||||||||||||||||
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
27 | ||||||||
2025 Activity
Research and Development Expenses
The increase in research and development expenses is mainly explained by the increase in costs related to GENFIT's product candidates, in particular VS-01, as well as increased staffing levels.
General and administrative expenses
The decrease in general and administrative employee expenses was mainly due to a decrease in related other expenses.
Marketing and market access expenses
Marketing and market access expenses remained stable period over period.
Employee expenses
Employee expenses and number of employees were as follows:
| Employee expenses | Half-year ended | ||||||||||||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | |||||||||||||||
| Wages and salaries | (7,848) | (8,401) | |||||||||||||||
| Social security costs | (3,025) | (3,361) | |||||||||||||||
| Changes in pension provision | (77) | (106) | |||||||||||||||
| Share-based compensation | (334) | (242) | |||||||||||||||
| TOTAL | (11,284) | (12,111) | |||||||||||||||
| Number of employees at year-end | Half-year ended | ||||||||||||||||
| 2024/06/30 | 2025/06/30 | ||||||||||||||||
| Average number of employees | 164 | 186 | |||||||||||||||
| Number of employees | |||||||||||||||||
| Research and development | 86 | 100 | |||||||||||||||
| Services related to research and development | 20 | 22 | |||||||||||||||
| Administration and management | 61 | 64 | |||||||||||||||
| Marketing and commercial | 2 | 2 | |||||||||||||||
| TOTAL | 169 | 188 | |||||||||||||||
The increase in employee expenses resulted mainly from an increase in workforce, with an average headcount from 164 in 2024 to 186 in 2025.
9.FINANCIAL INCOME AND EXPENSES
| Financial income and expenses | Half-year ended | |||||||||||||
| (in € thousands) | 2024/06/30 | 2025/06/30 | ||||||||||||
| Financial income | ||||||||||||||
| Interest income | 535 | 295 | ||||||||||||
| Foreign exchange gain | 271 | 322 | ||||||||||||
| Financial income from OCEANE repurchase | 0 | 326 | ||||||||||||
| Other financial income | 740 | 906 | ||||||||||||
| TOTAL - Financial income | 1,546 | 1,850 | ||||||||||||
| Financial expenses | ||||||||||||||
| Interest expenses | (2,327) | (1,126) | ||||||||||||
| Interest expenses for leases | (33) | (27) | ||||||||||||
| Royalty financing debt issuance costs | 0 | (4,020) | ||||||||||||
| Changes in fair value through profit or loss | 0 | (5,434) | ||||||||||||
| Foreign exchange losses | (42) | (1,399) | ||||||||||||
| Other financial expenses | (18) | (21) | ||||||||||||
| TOTAL - Financial expenses | (2,419) | (12,027) | ||||||||||||
| FINANCIAL GAIN (LOSS) | (873) | (10,178) | ||||||||||||
For further information related to the Royalty Financing issuance costs and changes in fair value of the Royalty Financing liability, see Note 17 - Loans and borrowings.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
28 | ||||||||
10.INCOME TAX
Tax expense
The income tax expense for the interim period is determined by applying management's best estimate of the weighted average tax rate for the annual period, adjusted for certain items fully applicable to the interim period if necessary to profit or loss before tax. The Group has made this assessment based on currently available information regarding the activities of the Group's entities, as well as the effective tax rates applicable in each relevant jurisdiction and historical data.
11.EARNINGS (LOSS) PER SHARE
Basic earnings (loss) per share are calculated by dividing profit or loss attributable to the Company's ordinary shareholders by the weighted average number of ordinary shares outstanding during the period.
Diluted earnings (loss) per share are calculated by adjusting profit attributable to ordinary shareholders and the average number of ordinary shares outstanding weighted for the effects of all potentially dilutive instruments (share warrants, redeemable share warrants, free shares, stock options and bonds convertible into new and/or existing shares).
The components of the earnings (loss) per share computation are as follows:
| Earnings per share | Half-year ended | |||||||||||||
| 2024/06/30 | 2025/06/30 | |||||||||||||
| Profit (loss) for the period (in € thousands) | 30,311 | (9,956) | ||||||||||||
| Basic earnings (loss) per share (€/share) | 0.61 | (0.20) | ||||||||||||
| Weighted average number of ordinary shares used to calculate diluted earnings (loss) per share | 60,363,017 | 49,784,805 | ||||||||||||
| Diluted earnings (loss) per share (€/share) | 0.53 | (0.20) | ||||||||||||
The weighted average numbers of ordinary shares as noted above exclude treasury shares held by GENFIT.
The following table summarizes the potential common shares not included in the computation of diluted earnings per share because their impact would have been antidilutive:
| Potential common shares not included in the computation of diluted earnings per share | Half-year ended | ||||||||||
| 2024/06/30 | 2025/06/30 | ||||||||||
| BSA | 0 | 0 | |||||||||
| STOCK OPTIONS | 0 | 1,314,650 | |||||||||
| AGA | 0 | 229,600 | |||||||||
| OCEANES | 0 | 108,939 | |||||||||
12.CASH AND CASH EQUIVALENTS
The main components of cash equivalents were:
•UCITS and interest-bearing current accounts, available immediately;
•Term accounts, available within the contractual maturities or by the way of early exit with no penalty; and
•Negotiable medium-term notes, available with a quarterly maturity or by the way of early exit with no penalty.
These investments, summarized in the tables below, are short-term, highly liquid and subject to insignificant risk of changes in value.
| Cash and cash equivalents | As of | |||||||||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||||||||
| Short-term deposits | 63,027 | 96,422 | ||||||||||||
| Cash on hand and bank accounts | 18,761 | 11,090 | ||||||||||||
| TOTAL | 81,788 | 107,511 | ||||||||||||
Short-term deposits are comprised of:
| Short-term deposits | As of | |||||||||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||||||||
| TERM ACCOUNTS | 63,027 | 96,422 | ||||||||||||
| TOTAL | 63,027 | 96,422 | ||||||||||||
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
29 | ||||||||
13.CURRENT TRADE AND OTHER RECEIVABLES
Financial statement line item detail
| Trade and other receivables - Total | As of | |||||||||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||||||||
| Trade receivables, net | 2,140 | 36,870 | ||||||||||||
| Research tax credit | 3,392 | 5,330 | ||||||||||||
| Social security costs receivables | — | 3 | ||||||||||||
| VAT receivables | 1,043 | 1,464 | ||||||||||||
| Grants receivables | 8 | 5 | ||||||||||||
| Other receivables | 981 | 38 | ||||||||||||
| TOTAL | 7,564 | 43,709 | ||||||||||||
| Of which : Current | 7,564 | 43,709 | ||||||||||||
| Of which : Non-current | — | — | ||||||||||||
Trade receivables, net
Trade receivables amounted to €36,870 as of June 30, 2025 and €2,140 as of December 31, 2024. The balance mainly corresponds to revenue related to the Licence and Collaboration Agreement with Ipsen.
Research tax credit
The research tax credit receivable as of June 30, 2025 amounts to €5,330 and €3,392 as of December 31, 2024.
VAT receivables
The VAT receivable amounted to €1,464 at June 30, 2025 (€1,043 at December 31, 2024).
Other receivables
The line item “other receivables” primarily consists of credit notes from suppliers as of June 30, 2025 and December 31, 2024.
14.OTHER CURRENT ASSETS
Other current assets amount to €4,204 at June 30, 2025 and €3,409 at December 31, 2024, respectively, and consisted of prepaid expenses related to current operating expenses.
15.INTANGIBLE ASSETS
Financial statement line item detail
| As of | Increase | Decrease | Translation | Reclassification | As of | |||||||||||||||||||||||||||||||||
| (in € thousands) | 2023/12/31 | adjustments | 2024/12/31 | |||||||||||||||||||||||||||||||||||
| Gross | ||||||||||||||||||||||||||||||||||||||
| Software | 955 | — | (180) | — | — | 776 | ||||||||||||||||||||||||||||||||
| Patents | 369 | — | — | — | (5) | 364 | ||||||||||||||||||||||||||||||||
| Other intangibles | 48,366 | — | — | (748) | — | 47,618 | ||||||||||||||||||||||||||||||||
| TOTAL—Gross | 49,690 | — | (180) | (748) | (5) | 48,757 | ||||||||||||||||||||||||||||||||
| Accumulated depreciation and impairment | ||||||||||||||||||||||||||||||||||||||
| Software | (928) | (10) | 180 | — | — | (759) | ||||||||||||||||||||||||||||||||
| Patents | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||
| Other intangibles | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||
| TOTAL - Accumulated depreciation and impairment | (928) | (10) | 180 | — | — | (759) | ||||||||||||||||||||||||||||||||
| TOTAL - Net | 48,761 | (10) | — | (748) | (5) | 47,998 | ||||||||||||||||||||||||||||||||
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
30 | ||||||||
| As of | Increase | Decrease | Translation | Reclassification | As of | |||||||||||||||||||||||||||||||||
| (in € thousands) | 2024/12/31 | adjustments | 2025/06/30 | |||||||||||||||||||||||||||||||||||
| Gross | ||||||||||||||||||||||||||||||||||||||
| Software | 776 | 34 | (165) | — | — | 645 | ||||||||||||||||||||||||||||||||
| Patents | 364 | — | — | — | 2 | 366 | ||||||||||||||||||||||||||||||||
| Other intangibles | 47,618 | 2,000 | — | 317 | — | 49,935 | ||||||||||||||||||||||||||||||||
| TOTAL—Gross | 48,757 | 2,034 | (165) | 317 | 2 | 50,945 | ||||||||||||||||||||||||||||||||
| Accumulated depreciation and impairment | ||||||||||||||||||||||||||||||||||||||
| Software | (759) | (5) | 165 | — | — | (599) | ||||||||||||||||||||||||||||||||
| Patents | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||
| Other intangibles | — | — | — | — | — | — | ||||||||||||||||||||||||||||||||
| TOTAL - Accumulated depreciation and impairment | (759) | (5) | 165 | — | — | (599) | ||||||||||||||||||||||||||||||||
| TOTAL - Net | 47,998 | 2,029 | — | 317 | 2 | 50,346 | ||||||||||||||||||||||||||||||||
Software
Software is comprised of office and scientific software.
Other intangibles
Other Intangibles assets consist of licensing agreements with Seal Rock and Celloram as well as technology acquired from Versantis and Genoscience Pharma. Each are detailed below.
Genoscience Pharma asset transfer (2025)
On January 3, 2025, GENFIT acquired all patents and patent applications, know-how, and data held by Genoscience Pharma necessary for the development, manufacturing, and marketing of GNS561, regardless of its therapeutic indication, form, dosage, or formulation. The sale price was a lump sum payment of €2 million euros excluding taxes. This agreement terminates the previous agreement signed in 2021.
In accordance with IAS 38 - Intangible assets this amount was capitalized and allocated to Intangible assets. Further, given the nature of the intangible asset, it was determined to have a definite useful life of 20 years, consistent with patent lifetimes in the United States and the European Union. Amortization will start upon EMA/FDA regulatory approval and until then will be subject to an annual impairment test in accordance with IAS 36 - Impairment of Assets. The annual impairment test will be based on a valuation methodology including an income approach using discounted cash flow techniques for GNS561 in Cholangiocarcinoma.
Indicators of impairment considered by the Group are as follows:
•Failure of or unfavorable data from our clinical trials
•Competition from other clinical trial programs covering the same indications as our drug candidates
•Availability of necessary financing
The value of the asset is €2 million at June 30, 2025. In 2025, there has been no indication of impairment.
Seal Rock license agreement (2023)
On May 31, 2023, GENFIT announced the signing of a licensing agreement for the exclusive worldwide rights to the injectable formulation of ASK1 inhibitor SRT-015 in acute liver disease with Seal Rock Therapeutics, a clinical-stage company based in Seattle, USA. Under the terms of the agreement, GENFIT made an upfront payment in the amount of €2 million to Seal Rock in exchange for acquiring the know-how and rights of use to SRT-015.
In accordance with IAS 38 - Intangible assets this amount was capitalized and allocated to Intangible assets. Further, given the nature of the intangible asset, it was determined to have a definite useful life of 20 years, consistent with patent lifetimes in the United States and the European Union. Amortization will start upon EMA/FDA regulatory approval and until then will be subject to an annual impairment test in accordance with IAS 36 - Impairment of Assets. As future milestones for this agreement are paid, they will be analyzed and be either i) capitalized and subject to the same annual impairment test or ii) expensed as incurred. The annual impairment test will be based on a valuation methodology including an income approach using discounted cash flow techniques for the injectable formulation of ASK1 inhibitor SRT-015 in acute liver disease.
Indicators of impairment considered by the Group are as follows:
•Failure of or unfavorable data from our clinical trials
•Competition from other clinical trial programs covering the same indications as our drug candidates
•Availability of necessary financing
The value of the asset is €2 million at June 30, 2025. In 2025, there has been no indication of impairment.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
31 | ||||||||
Versantis acquisition (VS-01) (2022)
On September 29, 2022, GENFIT acquired Versantis AG, a private Swiss-based clinical stage biotechnology company focused on addressing the growing unmet medical needs in liver diseases. The Phase 2 ready program, VS-01-ACLF, a program in scavenging liposomes technology, was deemed to be the asset with substantially all attributable value in accordance with the optional concentration test of fair value under paragraph B7A of IFRS 3.
Of the total acquisition price paid of €46.6 million, €43.9 million was allocated to Intangible assets. The difference between that amount and the acquisition price corresponds to the other assets acquired and liabilities assumed as part of the transaction. Further, given the nature of the intangible asset, it was determined to have a definite useful life of 20 years, consistent with patents lifetimes in the United States and the European Union. Amortization will start upon EMA/FDA regulatory approval and until then will be subject to an annual impairment test in accordance with IAS 38 - Intangible Assets.
Indicators of impairment considered by the Group are as follows:
•Failure of or unfavorable data from our clinical trials
•Competition from other clinical trial programs covering the same indications as our drug candidates
•Availability of necessary financing
The value of the asset is €45.8 million (after CHF/EUR currency translation adjustments) at June 30, 2025. In 2025, there has been no indication of impairment.
Adverse event related to the Versantis VS-01 intangible
On September 19, 2025, GENFIT announced its decision to discontinue its VS-01 program in ACLF (Acute-on-Chronic Liver Failure), and reprioritize its development on UCD (Urea Cycle Disorder). In accordance with IAS 10, this is considered on a non-adjusting event as this is a situation arising entirely after the reporting period. Refer to Note 2.2 - Events after the period.
Goodwill
The company does not have any goodwill.
16.OTHER NON-CURRENT FINANCIAL ASSETS
Other financial assets consisted of the following:
| Financial assets - Variations | As of | Increase | Decrease | As of | ||||||||||||||||||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||||||||||||||||||||
| Non consolidated equity investments | 1,425 | — | (114) | 1,311 | ||||||||||||||||||||||
| Other investments | 459 | — | (63) | 396 | ||||||||||||||||||||||
| Loans | 524 | 32 | — | 556 | ||||||||||||||||||||||
| Deposits and guarantees | 303 | 68 | (30) | 341 | ||||||||||||||||||||||
| Liquidity contract | 354 | 45 | — | 398 | ||||||||||||||||||||||
| TOTAL | 3,065 | 144 | (207) | 3,002 | ||||||||||||||||||||||
Non-consolidated equity investments
As of June 30, 2025, the value of "Non-consolidated equity investments" relates solely to our equity purchase in Genoscience Pharma. Since the transaction occurred, no shares have been sold. The gross value of the investment (and the initial transaction amount from 2021) totals €3,133. The net value of the investment (net of impairments) totals €1,311 as of June 30, 2025.
We did not complete the equity purchase in Genoscience Pharma for trading purposes. Therefore, pursuant to IFRS 9 and IAS 36, we elected to classify the equity in Genoscience Pharma we acquired in December 2021 as equity instruments recognized at fair value through other comprehensive income (OCI).
In accordance with IFRS 13, we updated our estimated of the fair value of our equity stake in Genoscience Pharma, which was based on a valuation methodology including a royalty based income approach using discounted cash flow techniques for the company's main scientific research programs. The aforementioned income method utilizes management’s estimates of future operating results, cash flows discounted using a weighted-average cost of capital that reflects market participant assumptions, and the expected success rate of each program.
Based on our analysis performed as of June 30, 2025, a loss of €114 was recorded in OCI.
The period over which management has projected its cash flows spans through 2038. The drug price growth rate used to extrapolate cash flow projections is 1%. Furthermore, we have performed the following sensitivity analyses in order to determine the change in value of the asset by modifying certain key assumptions.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
32 | ||||||||
Values assigned to each key assumption
•Discount rate: 12.5%
The amount by which the asset would decrease if the weighted average cost of capital increased by 1%: €109
•Overall expected success rate: 14.5%
The amount by which the asset would decrease if the estimated overall success rate decreased by 1%: €85
Indicators of impairment considered by the Group as part of the impairment test above are as follows:
•Failure of or unfavorable data from our clinical trials
•Competition from other clinical trial programs covering the same indications as our drug candidates
•Availability of necessary financing
Other investments
As of June 30, 2025, the value of "Other investments" totaled €396. The balance relates solely relates to our investment in CAPTECH SANTE.
Liquidity contract
Consistent with customary practice in the French securities market, we entered into a liquidity agreement ("Contrat de Liquidité") with Crédit Industriel et Commercial S.A. ("CIC") in August 2013. The liquidity agreement was entered into in accordance with applicable laws and regulations in France. The liquidity agreement authorizes CIC to carry out market purchases and sales of our shares on Euronext Paris.
As of June 30, 2025, the liquidity account had a cash balance of €398, and as of December 31, 2024 a cash balance of €354.
CIC holds the following number of GENFIT shares on behalf of the Company, recorded as a deduction in equity:
| Financial assets - Current | As of | ||||||||||||||||
| 2024/12/31 | 2025/06/30 | ||||||||||||||||
| Number of shares (recorded as a deduction from equity) | 207,500 | 201,100 | |||||||||||||||
17.LOANS AND BORROWINGS
17.1Royalty Financing
Non-Dilutive Royalty Financing Agreement
On January 30, 2025, GENFIT announced the signing of a royalty financing deal with HealthCare Royalty (HCRx) providing up to €185 million non-dilutive capital, including €130 million upfront, with eligibility to receive up to €55 million in two additional installments based on near-term milestones.
Installments
The royalty financing takes the form of an issuance by GENFIT of straight bonds to be subscribed by HCRx (the “Royalty Financing Bonds”), for an aggregate subscription price plus premium of up to €185 million (the “Subscription Price”, with a nominal value of €9.25 million). The Royalty Financing Bonds’ subscription price is arranged in up to three installments as follows:
•A first installment for a total subscription amount of €130 million, already issued on March 20, 2025 and received following i) approval of the OCEANEs bondholders at the bondholders meeting held on March 10, 2025 and ii) satisfaction of other customary closing conditions;
•A second installment for a total subscription amount of €30 million, subject to net sales of Iqirvo® (elafibranor) reaching a certain threshold by December 31, 2025; and
•A third installment for a total subscription amount of €25 million, subject to net sales of Iqirvo® (elafibranor) reaching a certain threshold by December 31, 2026.
Payment of the second and third installments are at the option of GENFIT, provided the corresponding conditions are met.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
33 | ||||||||
Repayment terms / multiples
The Royalty Financing Bonds issued by GENFIT will not bear interest. Instead, the returns on these bonds will be tied to a portion of the royalties GENFIT receives under the Ipsen agreement from October 1, 2024. This portion of the royalties is subject to the following multiples:
•If, at December 31, 2030, the cumulated amount of royalties received by HCRx represents 155% of the Subscription Price of the Royalty Financing Bonds, excluding the nominal value (i.e., approximately €277.5 million if all installments of the Subscription Price are paid), it will no longer be entitled to the royalties, which will, from then on, fully revert to the Company.
•If, at December 31, 2030, the cumulated amount of royalties received by HCRx represents less than this 155% return rate, it will continue to receive the royalties until the cumulated amount received represents 195% of the Subscription Price excluding the nominal value (i.e., up to a maximum of €351.5 million if all installments of the Subscription Price are paid).
•If, at December 31, 2033, this 195% return rate is not achieved, HCRx will continue to receive the royalties until the cumulated amount received equals 250% of the Subscription Price excluding the nominal value (i.e., up to a maximum of €453.25 million if all installments of the Subscription Price are paid).
Caps and time-limits
This portion of the royalties is subject to the following caps and time-limits:
•An annual cap equal to the amount of royalties based on an annual maximum amount of net sales of $600 million. The Company will receive 100% of the royalties based on the annual net sales exceeding this maximum.
•A final time-limit corresponding to the earlier of the following two dates: (i) the date on which the Company would no longer be entitled to receive royalties under the Ipsen agreement, and (ii) March 31, 2045 (notwithstanding the fact that none of the above return rates would have been achieved).
Nominal value repayment / HCRx recourse
When either i) the applicable multiple is fully repaid (excluding the nominal value) or ii) one of the above final time limits is reached, then the Company must repay the nominal amount of the Royalty Financing Bonds (i.e. €9.25 million).
HCRx’s recourse against GENFIT is limited to GENFIT’s non-compliance with its contractual obligations under the royalty financing documentation and repayment of the nominal value of the Royalty Financing Bonds (€9.25 million).
French law trust (fiducie-sûreté)
To secure its payment and repayment obligations under the Royalty Financing Bonds, GENFIT will transfer the corresponding royalty receivables to a French law trust (fiducie-sûreté) for the benefit of the holders of the royalty financing bonds. To grant the security interest on the royalty receivable, the Company obtained the consent of the OCEANEs holders.
Royalty Financing accounting, balances and disclosures
Initial accounting
The first installment of €130 million was received on March 20, 2025. Therefore, GENFIT has designated the Royalty Financing is accounted for as a liability that includes an inseparable derivative and, in accordance with IFRS 9, is measured at fair value.
The total amount of the liability depends on royalties received from Ipsen, which are determined based on the sales volume of products derived from the licensed molecule—currently the drug Iqirvo® (elafibranor)—and denominated in the sales currency, which is predominantly U.S. dollars. Although these royalties can be estimated, they remain variable, introducing the concept of an underlying that can affect the value of the liability—a typical characteristic of derivatives. Furthermore, the royalties owed by Ipsen to Genfit are payable exclusively in euros, based on the average exchange rate over the 30-day period preceding the royalty payment due date. As a result, the repayment flows of the obligation depend on the evolution of the EUR/USD exchange rate, particularly in relation to the annual net sales cap of USD 600 million.
Under IFRS 9, to determine whether the embedded derivative should be accounted for separately from the host contract, one must assess whether the economic characteristics and risks of the embedded derivative are closely related to those of the host contract. In this case, the reliance on the revenue of a third party (Ipsen) introduces a commercial risk that is exogenous to the typical credit risk of a debt instrument. This risk is not closely related to a debt whose cash flows are normally fixed or indexed to “standard” financial parameters. As a result, in this case, the embedded derivative cannot be considered closely related to the host contract.
Key initial estimates
GENFIT plans to fulfill its commitment to HCRx before December 31, 2030. Accordingly, GENFIT will recognize the difference between the net proceeds received (€130 million) and the total expected payments (€201.2 million) as fair value through profit or loss (FVTPL) expenses.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
34 | ||||||||
Key assumptions used in the fair value assessment are as follows:
•The expected sales of Iqirvo® (elafibranor) in multiple jurisdictions
•The forecasted exchange rates applicable to sales denominated in currencies other than the euro (primarily U.S. dollars), and
•The expected discount rate (which is nonobservable input).
These assumptions will be reviewed and updated at each reporting date. Changes in estimates will be recognized through fair value-related expenses in profit or loss (FVTPL).
As of June 30, 2025, a simplified amortized cost model was used to estimate the fair value of the Royalty Financing, taking into account the aforementioned assumptions, given the time elapsed between the start of the contract (March 20, 2025) and the reporting date (June 30, 2025). In accordance with IAS 1, we applied reasonable assumptions to establish accounting estimates, considering the cost-benefit. After June 30, 2025, GENFIT expects to utilize a stochastic model, a mathematical tool that incorporates uncertainty and variability of random processes, which is generally used to value embedded derivatives.
Note on debt issuance costs
Total debt issuance costs incurred totalled €4.0 million and were recognized in "Financial expenses" in the Consolidated statement of operations in the first half of 2025.
Under the agreement, HCRx incurred and paid an amount of €424 thousand in eligible expenses. In accordance with the terms of the agreement, and at its discretion and independently of any other outcome related to the agreement, GENFIT is obligated to reimburse HCRx for these expenses, inclusive of the applicable contractual multiple (as indicated above), depending on the timing of the reimbursement. Accordingly, GENFIT concluded that this additional liability will be repaid before December 31, 2030, for a total amount of €657 thousand (€424 thousand inclusive of the applicable 155% multiple). As a result, the amount recognized in profit or loss on March 20, 2025, was €657 thousand, and the balance will be settled before December 31, 2030. As of June 30, 2025, this amount is presented under 'Liability from royalty financing agreement' on the Statement of Financial Position.
Activity and balances
The following table presents the movement in the liability related to the Royalty Financing agreement between March 20, 2025 and June 30, 2025:
| Amount of liability | |||||
| (in € thousands) | |||||
Gross proceeds on March 20, 2025 | 130,000 | ||||
Royalty payments to HCRx via the French-law trust | (4,492) | ||||
Unpaid issuance costs | 657 | ||||
| Fair value through profit or loss (FVTPL) expenses | 5,434 | ||||
Royalty financing agreement liability as of June 30, 2025 | 131,599 | ||||
Expected future cash flows
The following royalty revenues expected to be used to repay the Royalty Financing agreement are as follows:
| Year | Royalties (in € millions) | ||||
2025 (remainder thereof) | 7.0 | ||||
2026 | 26.2 | ||||
2027 | 46.2 | ||||
2028 | 58.7 | ||||
2029 | 58.9 | ||||
Sensitivity
Forecast USD/EUR exchange rate
The Group expects to be able to fully reimburse the first installment of the Royalty financing agreement prior to the end of 2029 based on expected future cash flows and thus fulfill reimbursement at the contractual multiple of 155%. The estimated average USD/EUR exchange rate is 1.08 (1 EUR = 1.08 USD). If, for this first installment, the actual average exchange rate over the entire repayment period were 1.70 (with the underlying assumption that US sales make up 70% of total sales), this would cause the expected reimbursement to occur after 2030 and thus would trigger the application of the contractual multiple of 195%.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
35 | ||||||||
Future sales
The Group expects to be able to fully reimburse the first installment of the Royalty financing agreement prior to the end of 2029 based on expected future cash flows and thus fulfill reimbursement at the contractual multiple of 155%. If, on the first installment, future cash flows decrease on average by greater than 31% this would cause the expected reimbursement to occur after 2030 and thus would trigger the application of the contractual multiple of 195%.
17.2.Breakdown of convertible loan
On October 16, 2017, the Company issued 6,081,081 OCEANEs at par with a nominal unit value of €29.60 per bond for an aggregate nominal amount of €180 million. This debt was renegotiated in January 2021 and substantially repurchased in 2025.
Updated nominal balances
| As of 31/12/2024 : | ||||||||
| Number of bonds | 1,902,698 | |||||||
| Nominal amount of the loan | 56,319,860.80€ | |||||||
| Nominal unit value of the bonds | 29.60€ | |||||||
| Effective interest rate | 8.8% | |||||||
| As of 30/06/2025 : | ||||||||
| Number of bonds | 19,807 | |||||||
| Nominal amount of the loan | 586,287.20€ | |||||||
| Nominal unit value of the bonds | 29.60€ | |||||||
| Effective interest rate | 8.8% | |||||||
Repurchase
Currently with the Royalty Financing agreement (see Note 17.1 - Royalty Financing), the Company proposed to all of the OCEANEs holders to enter into a Put Option Agreement, pursuant to which the Company unconditionally and irrevocably undertook to repurchase the OCEANEs of such holder at a price of EUR 32.75 per bond, subject to approval by the general meeting of the OCEANEs holders of the amendment of the terms and conditions of the OCEANEs and the closing of the Royalty Financing (the “Repurchase”). Holders had until March 19, 2025 to exercise this option.
The settlement of the Repurchase occurred on March 26, 2025. 1,882,891 OCEANEs were repurchased for a total amount paid of €61.7 million. The repurchased OCEANEs were then canceled by the Company. A new calculation of the “debt” and “equity” components was performed on the repurchase date, taking into account the updated discount rate as of that date, in accordance with IAS 32. This allowed the repurchase price to be allocated between the “debt” component (€54.8 million) and the “equity” component (€6.9 million).
Just prior to the repurchase, the net carrying amount of the “debt” component of the repurchased OCEANEs was €55.1 million. As a result, a gain of €0.3 million was recognized in Financial Income on the Consolidated Statement of Operations, corresponding to the difference between the repurchase price allocated to the “debt” component (€54.8 million) and its carrying amount (€55.1 million).
The portion of the repurchase price allocated to the “equity” component (€6.9 million) was recorded as a deduction from Share Premium in the Consolidated Statement of Changes in Equity.
Consent Fee
The Company also undertook, subject to the approval of the amendment of the terms and conditions of the OCEANEs and the closing of the Royalty Financing, to pay a consent fee (the “Consent Fee”) of EUR 0.90 per bond to the holders of OCEANEs still outstanding after cancellation of the repurchased OCEANEs. The OCEANEs that were bought back by the Company as part of the Repurchase thus did not receive the Consent Fee.
The payment of the Consent Fee occurred on April 14, 2025, totaling €18 thousand recorded in Financial expenses.
Key terms and other information
Nominal annual interest rate
The nominal annual interest rate is 3.5%, payable semi-annually in arrears.
Repayment terms
Final reimbursement is scheduled for October 16, 2025.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
36 | ||||||||
Redemption prior to maturity at the option of the Company is possible if the arithmetic volume-weighted average price of GENFIT's listed share price and the then prevailing conversion ratio over a 20 day trading period exceeds 1.5 times the nominal value of the OCEANEs.
Conversion ratio and terms
The conversion ration is 5.5 ordinary shares per bond. There are no specific terms that need to be met for a holder of OCEANEs to convert their debt into GENFIT shares.
50 bonds were requested to be converted into 275 GENFIT shares during the six month period ended June 30, 2025 after the repurchase described above (finalized and recorded in July).
Conversion / exchange premium
The conversion / exchange premium is 30% relative to GENFIT's reference share price (22.77€).
Maximum dilution
The potential issuance of new shares upon conversion requests of the outstanding OCEANEs would represent 0.2% of the share capital of the Company at June 30, 2025.
Remaining balances
| Convertible loans - Current | As of | |||||||||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||||||||
| Convertible loans | 54,572 | 582 | ||||||||||||
| TOTAL | 54,572 | 582 | ||||||||||||
17.3.Breakdown of other loans and borrowings
Other loans and borrowings consisted of the following:
| Other loans and borrowings - Total | As of | |||||||||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||||||||
| Bank loans | 2,496 | 2,076 | ||||||||||||
| Obligations under leases | 5,060 | 4,652 | ||||||||||||
| Accrued interests | 5 | 4 | ||||||||||||
| TOTAL | 7,561 | 6,732 | ||||||||||||
| Other loans and borrowings - Current | As of | |||||||||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||||||||
| Bank loans | 859 | 858 | ||||||||||||
| Obligations under leases | 1,145 | 1,182 | ||||||||||||
| Accrued interests | 5 | 4 | ||||||||||||
| TOTAL | 2,009 | 2,044 | ||||||||||||
| Other loans and borrowings - Non current | As of | |||||||||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||||||||
| Bank loans | 1,637 | 1,218 | ||||||||||||
| Obligations under leases | 3,915 | 3,470 | ||||||||||||
| Accrued interests | — | — | ||||||||||||
| TOTAL | 5,552 | 4,688 | ||||||||||||
Bank Loans
See Note 20.2.2 "Bank loans" in the Notes to the Consolidated Financial Statements in the Company's 2024 20-F filing for a detailed description of the Group's bank loans and related accounting treatment.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
37 | ||||||||
Balances by loan
Bank loans consisted of the following as of December 31, 2024 and June 30, 2025:
| Bank loans | Loan | Facility | Interest | Available As of 2025/06/30 | Installments | Outstanding As of 2024/12/31 | Outstanding As of 2025/06/30 | |||||||||||||||||||||||||||||||||||||
| (in € thousands) | date | size | rate | |||||||||||||||||||||||||||||||||||||||||
| AUTRES | - | — | — | % | — | — | 9 | 19 | ||||||||||||||||||||||||||||||||||||
| BPI PGE | July 2021 | 2,000 | 2.25 | % | — | 16 quarterly | 1,100 | 900 | ||||||||||||||||||||||||||||||||||||
| BPI PRÊT TAUX BONIFIE | November 2021 | 2,250 | 2.25 | % | — | 20 quarterly | 1,380 | 1,157 | ||||||||||||||||||||||||||||||||||||
| TOTAL | 2,489 | 2,076 | ||||||||||||||||||||||||||||||||||||||||||
17.4Maturities of financial liabilities
| Maturity of financial liabilities | As of | Less than | Less than | Less than | Less than | Less than | More than | |||||||||||||||||||||||||||||||||||||
| (in € thousands) | 2025/06/30 | 1 year | 2 years | 3 years | 4 years | 5 years | 5 years | |||||||||||||||||||||||||||||||||||||
| Convertible loans | 591 | 591 | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||
| Bank loans | 2,076 | 858 | 869 | 341 | 5 | 3 | — | |||||||||||||||||||||||||||||||||||||
| Leases | 4,652 | 1,182 | 1,196 | 1,209 | 1,065 | — | — | |||||||||||||||||||||||||||||||||||||
| Accrued interests | 4 | 4 | — | — | — | — | — | |||||||||||||||||||||||||||||||||||||
| TOTAL - Other loans and borrowings | 7,323 | 2,635 | 2,065 | 1,550 | 1,070 | 3 | — | |||||||||||||||||||||||||||||||||||||
| TOTAL | 7,323 | 2,635 | 2,065 | 1,550 | 1,070 | 3 | — | |||||||||||||||||||||||||||||||||||||
The values in the table above are nominal (contractual) values according to IFRS 7.39(a).
For detail on the expected future payments related to the Royalty Financing liability, refer to Note 17.1 - Royalty Financing.
17.5Reconciliation of financial liabilities arising from financing activities
| As of | Cash flows | Non cash changes | As of | |||||||||||
| 31/12/2024 | 6/30/2025 | |||||||||||||
Convertible loan | 54,572 | (61,665) | 7,675 | 582 | ||||||||||
| Bank loans | 2,496 | (420) | 2,076 | |||||||||||
Royalty Financing Agreement | — | 125,508 | 6,091 | 131,599 | ||||||||||
| Obligations under leases | 5,060 | (577) | 169 | 4,652 | ||||||||||
| TOTAL | 62,128 | 138,909 | ||||||||||||
Non cash changes related to the convertible loan stem from the 2025 repurchase as detailed in Note 17.2 - Breakdown of Convertible loan.
Non cash changes related to the Royalty Financing agreement is from changes in fair value.
Non cash changes related to leases stems solely to annual contractual price indexation increases.
18.CURRENT TRADE AND OTHER PAYABLES
Financial detail
Current trade and other payables consisted of the following:
| Trade and other payables - Total | As of | |||||||||||||
| (in € thousands) | 2024/12/31 | 2025/06/30 | ||||||||||||
| Trade payables | 13,437 | 14,010 | ||||||||||||
| Social security costs payables | 4,092 | 3,077 | ||||||||||||
| VAT payables | 351 | 6,288 | ||||||||||||
| Taxes payables | 218 | 176 | ||||||||||||
| Other payables | 289 | 205 | ||||||||||||
| TOTAL | 18,387 | 23,757 | ||||||||||||
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
38 | ||||||||
At June 30, 2025, trade payables amounted to €14,010 (€13,437 at December 31, 2024). This change is due to a reduction in accrued expenses relating to yet unbilled amounts from the clinical trial sites via the Clinical Research Organizations (CROs) in charge of the Company's clinical trials (€8,736 and €5,426 at June 30, 2025 and December 31, 2024 respectively). The timeframe in which those invoices will be received by the Company is unknown and may be spread out over a long period after the services have been performed.
19.CURRENT PROVISIONS
Financial detail
At June 30, 2025 and at December 31, 2024, this line item amounted to €40 and €40, respectively.
| Change in provisions | As of | Increase | Decrease | Decrease | As of | |||||||||||||||
| (in € thousands) | 2024/12/31 | (used) | (unused) | 2025/06/30 | ||||||||||||||||
| Provision for charges | 40 | — | 0 | 0 | 40 | |||||||||||||||
| TOTAL | 40 | — | 0 | 0 | 40 | |||||||||||||||
20.EQUITY
Detailed breakdown
Share capital
| Number of shares | As of | |||||||||||||
| 2024/12/31 | 2025/06/30 | |||||||||||||
| Ordinary shares issued (€0.25 par value per share) | 49,996,185 | 50,002,560 | ||||||||||||
| Convertible preferred shares registered | — | — | ||||||||||||
| Total shares issued | 49,996,185 | 50,002,560 | ||||||||||||
| Less treasury shares | — | — | ||||||||||||
| Outstanding shares | 49,996,185 | 50,002,560 | ||||||||||||
Ordinary shares are classified under shareholders' equity. Any shareholder, regardless of nationality, whose shares are fully paid-in and registered for at least two years, is entitled to double voting rights under the conditions prescribed by law (Article 32 of the Company's bylaws).
Changes in share capital in 2025
During the six month period ended June 30, 2025:
•6,375 stock options were converted to ordinary GENFIT shares.
At June 30, 2025, the remaining unused authorizations to issue additional share-based compensation or other share-based instruments (stock options, free shares and share warrants) represent a total of 725,000 shares.
Share premium
The repurchase of OCEANEs in the first semester of 2025 included the corresponding derecognition of the option component recognized in "Share premium" in equity of €6.9 million. Refer to Note 17.2 - Breakdown of convertible loan.
Currency translation adjustment
The currency translation differences arise from the application of IAS 21 when converting the functional currencies of the Group's subsidiaries (i.e. the US dollar for GENFIT Corp and the Swiss franc for Versantis AG) into euros at each closing. The change period over period stems from the change of these two currencies' foreign exchange rates against the euro.
21.FAIR VALUE OF FINANCIAL INSTRUMENTS
Financial detail
The following tables provide the financial assets and liabilities carrying values by category and fair values as of June 30, 2025 and December 31, 2024:
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
39 | ||||||||
| As of 31/12/2024 | |||||||||||||||||||||||||||||
| Carrying value | Fair value | ||||||||||||||||||||||||||||
| As per | Assets at | Assets at | Assets at | Debt at | Level 1 | Level 2 | Level 3 | ||||||||||||||||||||||
| statement of | fair value | fair value | amortized | amortized | |||||||||||||||||||||||||
| financial | through | through OCI | cost | cost | |||||||||||||||||||||||||
| (in € thousands) | position | profit & loss | |||||||||||||||||||||||||||
| Assets | |||||||||||||||||||||||||||||
| Equity investments | 1,425 | 1,425 | 1,425 | ||||||||||||||||||||||||||
| Other investments | 459 | 459 | 459 | ||||||||||||||||||||||||||
| Loans | 524 | 524 | 524 | ||||||||||||||||||||||||||
| Deposits and guarantees | 303 | 303 | 303 | ||||||||||||||||||||||||||
| Liquidity contracts | 354 | 354 | 354 | ||||||||||||||||||||||||||
| Trade receivables | 2,140 | 2,140 | 2,140 | ||||||||||||||||||||||||||
| Cash and cash equivalents | 81,788 | 81,788 | 81,788 | ||||||||||||||||||||||||||
| TOTAL - Assets | 86,993 | 82,601 | 1,425 | 2,967 | — | 82,142 | 2,967 | 1,885 | |||||||||||||||||||||
| Liabilities | |||||||||||||||||||||||||||||
| Convertible loans | 54,572 | 54,572 | 56,320 | ||||||||||||||||||||||||||
| Bank loans | 2,496 | 2,496 | 2,496 | ||||||||||||||||||||||||||
| Obligations under finance leases | 5,060 | 5,060 | 5,060 | ||||||||||||||||||||||||||
| Accrued interests | 5 | 5 | 5 | ||||||||||||||||||||||||||
| Trade payables | 13,437 | 13,437 | 13,437 | ||||||||||||||||||||||||||
| Other payables | 289 | 289 | 289 | ||||||||||||||||||||||||||
| TOTAL - Liabilities | 75,859 | — | — | — | 75,859 | — | 77,607 | — | |||||||||||||||||||||
| As of 30/06/2025 | |||||||||||||||||||||||||||||||||||||||||||||||
| Carrying value | Fair value | ||||||||||||||||||||||||||||||||||||||||||||||
| As per | Fair value | Fair value | Assets at | Debt at | Level 1 | Level 2 | Level 3 | ||||||||||||||||||||||||||||||||||||||||
| statement of | through | through OCI | amortized | amortized | |||||||||||||||||||||||||||||||||||||||||||
| financial | profit & loss | cost | cost | ||||||||||||||||||||||||||||||||||||||||||||
| (in € thousands) | position | ||||||||||||||||||||||||||||||||||||||||||||||
| Assets | |||||||||||||||||||||||||||||||||||||||||||||||
| Equity investments | 1,311 | 1,311 | 1,311 | ||||||||||||||||||||||||||||||||||||||||||||
| Other investments | 396 | 396 | 396 | ||||||||||||||||||||||||||||||||||||||||||||
| Loans | 556 | 556 | 556 | ||||||||||||||||||||||||||||||||||||||||||||
| Deposits and guarantees | 341 | 341 | 341 | ||||||||||||||||||||||||||||||||||||||||||||
| Liquidity contracts | 398 | 398 | 398 | ||||||||||||||||||||||||||||||||||||||||||||
| Trade receivables | 36,870 | 36,870 | 36,870 | ||||||||||||||||||||||||||||||||||||||||||||
| Cash and cash equivalents | 107,511 | 107,511 | 107,511 | ||||||||||||||||||||||||||||||||||||||||||||
| TOTAL - Assets | 147,384 | 108,306 | 1,311 | 37,766 | — | 107,910 | 37,766 | 1,707 | |||||||||||||||||||||||||||||||||||||||
| Liabilities | |||||||||||||||||||||||||||||||||||||||||||||||
| Convertible loans | 582 | 582 | 588 | ||||||||||||||||||||||||||||||||||||||||||||
| Bank loans | 2,076 | 2,076 | 2,076 | ||||||||||||||||||||||||||||||||||||||||||||
| Obligations under finance leases | 4,652 | 4,652 | 4,652 | ||||||||||||||||||||||||||||||||||||||||||||
| Accrued interests | 4 | 4 | 4 | ||||||||||||||||||||||||||||||||||||||||||||
| Trade payables | 14,010 | 14,010 | 14,010 | ||||||||||||||||||||||||||||||||||||||||||||
| Liability from royalty financing agreement | 131,599 | 131,599 | 131,599 | ||||||||||||||||||||||||||||||||||||||||||||
| Other payables | 205 | 205 | 205 | ||||||||||||||||||||||||||||||||||||||||||||
| TOTAL - Liabilities | 153,128 | 131,599 | — | — | 21,529 | — | 21,536 | 131,599 | |||||||||||||||||||||||||||||||||||||||
22.LITIGATION
Not applicable.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
40 | ||||||||
23.RELATED PARTIES
Biotech Avenir
Biotech Avenir SAS is a holding company incorporated in 2001 by the Company's founders. Most of its share capital is currently held by individuals, i.e. the four co-founders of the Company and twelve Company employees.
Jean-François Mouney, the Chairman of the Company, is also the Chairman of Biotech Avenir SAS.
At June 30, 2025, Biotech Avenir SAS held 3.79% of the share capital of the Company.
The Company did not carry out any transactions with Biotech Avenir in 2025 or 2024, with the exception of the domiciliation without charge.
Ipsen Pharma SAS
The licensing agreement signed with Ipsen Pharma SAS in December 2021 provides for a certain number of service agreements that were signed with the Company in 2022 and 2023, notably the Transition Services Agreement and the Part B Transition Services Agreement.
These agreements cover support for Ipsen in future proceedings and processes (other than knowledge transfer) and the provision of drug tablets which Ipsen may require to execute its clinical trial. As per the agreement signed with Ipsen in December 2021, the prices under these agreements cover all costs borne by the Company to provide the relevant goods and services, without economic benefit for Ipsen. See Note 24 - Commitments, contingent liabilities and contingent assets.
24.COMMITMENTS, CONTINGENT LIABILITIES AND CONTINGENT ASSETS
Commitments
Obligations under the terms of subcontracting agreements
The Group enters into contracts for its business needs with clinical research organizations (CROs) for clinical trials, as well as with Contract Manufacturing Organizations (CMOs) for clinical and commercial supply manufacturing, commercial and pre-commercial activities, research and development activities and other services and products for operating purposes. The Group’s agreements generally provide for termination with specified periods of advance notice.
Such agreements are generally cancellable contracts and not included in the description of the Group’s contractual obligations and commitments.
Obligations under the terms of lease agreements
The Company has guaranteed its rental payment obligation under the lease agreement for the headquarters in Loos, France in the amount of €600 at June 30, 2025.
Planned capital expenditures
Capital expenditures (Scientific and IT investments) for which the Group has already made firm commitments amount to €100 as of the date of this half-year financial report. The Group plans to finance these investments over the next 12 months with available cash or new borrowings.
In addition, the Company will evaluate any opportunity to acquire new molecules that may complement those in its existing portfolio. If such an opportunity were taken, the Group would therefore make significant investments in this regard in the years to come. As of the date of this half-year financial report, the Group has not made any commitments in this regard.
Contingent liabilities
Obligations under the terms of the asset transfer protocol with Genoscience
On December 10, 2024, in the context of a “conciliation” (a French law, amicable pre-insolvency proceeding), GENFIT and Genoscience Pharma entered into an asset transfer protocol which entered into legal force on January 3, 2025 and terminates the previous agreement signed in 2021. Under the terms of this agreement:
•Contingent liabilities
◦(Patent sales) Genoscience Pharma will receive 25% of the proceeds of the sale of one or several GNS561 patents actually received by GENFIT.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
41 | ||||||||
◦(Commercialization) Genoscience Pharma will receive 25% of the net profits actually collected by GENFIT corresponding to the sales of said products for a period expiring on the earliest of the following dates (i) 10 years from the first commercialized sale, (ii) the expiration, cancellation or revocation of one of the patents or (iii) the authorization of generic products utilizing the GNS Technology.
◦(Licensing-out) Genoscience Pharma will receive 25% of 1) any milestone payments (including upfront milestone payments) from GNS561 patent licensing and 2) royalties from said patent licensing actually collected by GENFIT during a period expiring on the earlier of (i) 10 years from the first commercialized sale, (ii) the expiration, cancellation or revocation of one of the patents or (iii) the authorization of generic products implementing the GNS Technology.
•Time limit: Such amounts would only be due if one of the above circumstances occur within five years of the date of signing the 2025 transfer protocol (December 10, 2024).
In accordance with IAS 37, our obligations under the terms of the agreement we entered into with Genoscience Pharma constitute contingent liabilities not recognized in the Company's consolidated financial statements at December 31, 2024 and June 30, 2025.
Obligations under the terms of license agreement with Seal Rock
On May 31, 2023, GENFIT announced the signing of a licensing agreement for the exclusive worldwide rights to the ASK1 inhibitor SRT-015 with Seal Rock Therapeutics, a clinical-stage company based in Seattle, Washington. Under the terms of this agreement:
•Seal Rock is eligible for payments of up to €100 million (of which €2 million was paid in 2023), subject to certain regulatory, clinical and commercial outcomes.
•Seal Rock is likewise eligible for tiered royalties, applied to the annual sales of licensed products realized by GENFIT.
The conditional payments will be subject to analysis when they are incurred to determine if they are eligible for capitalization in accordance with IAS 38. If so, they will be capitalized. Otherwise, they will be expensed as incurred.
These obligations constitute contingent liabilities not recognized in the Company's consolidated financial statements at December 31, 2024 and June 30, 2025.
Obligations related to the licensing agreement with Celloram
On July 28, 2023, GENFIT licensed the exclusive worldwide rights to CLM-022, a first-in-class inflammasome inhibitor, from Celloram Inc., a Cleveland-based biotechnology company. Under the terms of the agreement:
•Celloram is eligible for payments of up to €160 million (of which €50 thousand have been paid in 2023), subject to certain regulatory, clinical and commercial outcomes.
•Celloram is likewise eligible for tiered royalties, applied to the annual sales of licensed products realized by GENFIT.
The conditional payments will be subject to analysis when they are incurred to determine if they are eligible for capitalization in accordance with IAS 38. If so, they will be capitalized. Otherwise, they will be expensed as incurred.
These obligations constitute contingent liabilities not recognized in the Company's consolidated financial statements at December 31, 2024 and June 30, 2025.
Obligations related to the Versantis acquisition
On September 29, 2022, GENFIT finalized an exclusive agreement to acquire all of the shares and voting rights of Versantis, a privately held clinical-stage biotechnology company based in Switzerland focused on addressing the growing medical needs in the field of liver diseases.
Under the terms of this agreement:
•The former shareholders of Versantis are eligible to receive certain payments of up to 65 million CHF, contingent on the following outcomes:
◦positive Phase 2 results related to VS-01-ACLF,
◦regulatory approval of VS-01-ACLF, and
◦positive Phase 2 results related to VS-02.
•Furthermore, the former shareholders of Versantis are eligible to receive 1/3 of the net proceeds resulting from the potential sale of the Priority Review Voucher of VS-01’s pediatric application by GENFIT to a third party, or 1/3 of the fair market value of this Voucher if GENFIT opts to apply it to one of its own programs.
The conditional payments will be subject to analysis when they are incurred to determine if they are eligible for capitalization in accordance with IAS 38. If so, they will be capitalized. Otherwise, they will be expensed as incurred.
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
42 | ||||||||
These obligations constitute contingent liabilities not recognized in the Company's consolidated financial statements at December 31, 2024 and June 30, 2025.
Adverse event related to the Versantis VS-01 intangible
On September 19, 2025, GENFIT announced its decision to discontinue its VS-01 program in ACLF (Acute-on-Chronic Liver Failure), and reprioritize its development on UCD (Urea Cycle Disorder). In accordance with IAS 10, this is considered on a non-adjusting event as this is a situation arising entirely after the reporting period. Refer to Note 2.2 - Events after the period.
Contingent assets
Contingent assets related to the licensing agreement with IPSEN
In December 2021, GENFIT and Ipsen Pharma SAS ("Ipsen") entered into an exclusive worldwide licensing agreement (except for China, Hong Kong, Taiwan and Macao, which apply to Terns as noted below) for elafibranor, a Phase 3 asset evaluated in Primary Biliary Cholangitis (PBC), as part of a long-term global partnership ("Collaboration and License Agreement"). Under this agreement we could receive milestone payments based on future events that are uncertain and therefore they constitute contingent assets not recognized in the Company's consolidated financial statements for the period ending June 30, 2025.
•GENFIT is also eligible for total milestone payments up to €360 million. These milestone payments constitute future variable income, dependent on the completion of key steps related to the development and sales of the licensed products. As such, in accordance with IFRS 15, this income will be recognized as revenue depending on the completion of these milestones. GENFIT has recognized cumulative milestones of €88.5 million as of June 30, 2025.
•GENFIT is eligible for tiered double-digit royalties of up to 20%, applied to the annual sales of licensed products realized by Ipsen. As such, in accordance with IFRS 15, this income will be recognized as revenue depending on the realization of these sales. Refer to Note 7 - Revenues and other income.
Contingent assets related to the licensing agreement with Terns Pharma
The Company entered into a licensing agreement with Terns Pharma whereby we could receive milestone payments based on future events that are uncertain and therefore they constitute contingent assets not recognized in the Company's consolidated financial statements for the period ending June 30, 2025. The licensing agreement with Terns concerns China, Hong Kong, Taiwan and Macao.
Milestones include Development Milestone Payments upon the achievement of the development milestones for the licensed product and Commercial Milestone Payments upon the achievement of commercial milestones depending on reaching certain aggregate thresholds. There are also potential mid-teen royalties based on sales by Terns Pharmaceuticals in Greater China. The potential Development and Commercial Milestone payments may represent up to $193 million.
25.SUPPLEMENTAL CASH FLOW INFORMATION
Supplemental cash flow information
Disclosure of non-cash financing and investing activities
•Accrued property, plant and equipment, at December 31, 2024: €196
•Accrued property, plant and equipment, at June 30, 2025: €208
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
43 | ||||||||
| 4. | STATUTORY AUDITORS’ LIMITED REVIEW REPORT ON 2025 HALF-YEAR CONDENSED CONSOLIDATED FINANCIAL STATEMENTS | |||||||
| GRANT THORNTON | ERNST & YOUNG et Autres | ||||
Membre français de Grant Thornton International 29, rue du Pont 92200 Neuilly-sur-Seine S.A.S. au capital de € 2 297 184 632 013 843 R.C.S. Nanterre Commissaire aux Comptes Membre de la compagnie régionale de Versailles et du Centre | Tour First, TSA 14444 92037 Paris-La Défese cedex S.A.S. à capital variable 438 476 913 R.C.S. Nanterre Commissaire aux Comptes Membre de la compagnie régionale de Versailles et du Centre | ||||
GENFIT
For the period from 1 January to 30 June 2025
Statutory auditors’ review report on the half-yearly financial information
To the Shareholders,
In compliance with the assignment entrusted to us by your Annual General Meeting and in accordance with the requirements of article L. 451-1-2 III of the French Monetary and Financial Code (Code monétaire et financier), we hereby report to you on:
•.the review of the accompanying condensed half-yearly consolidated financial statements of GENFIT, for the period from 1 January to 30 June 2025;
•.the verification of the information presented in the half-yearly management report.
These condensed half-yearly consolidated financial statements were drawn up under the responsibility of the Board of Directors. Our role is to express a conclusion on these financial statements based on our review.
1. Conclusion on the financial statements
We conducted our review in accordance with the professional standards applicable in France.
A limited review of interim financial information consists of making inquiries of persons responsible for financial and accounting matters, and of applying analytical procedures. This review is substantially less in scope than an audit conducted in accordance with the professional standards applicable in France and consequently does not enable us to obtain assurance that we would become aware of all significant matters that might be identified in an audit. Accordingly, we do not express an audit opinion.
Based on our limited review, nothing has come to our attention that causes us to believe that the accompanying condensed half-yearly consolidated financial statements are not prepared, in all material respects, in accordance with standard IAS 34 of the IFRS as adopted by the European Union applicable to interim financial information.
Without modifying our conclusion, we draw your attention to the matter set out in note 2.2 “Events after the period” to the condensed half-year consolidated financial statements, which describes a major adverse event related to the intangible asset Versantis VS-01 that occurred in September 2025.
2. Specific verification
We have also verified the information presented in the half-yearly management report on the condensed half-yearly consolidated financial statements subject to our limited review.
We have no matters to report as to its fair presentation and consistency with the condensed half-yearly consolidated financial statements.
Neuilly-sur-Seine and Paris-La Défense, 22 September 2025
The Statutory Auditors
(French original signed by)
GRANT THORNTON Membre français de Grant Thornton International | ERNST & YOUNG et Autres | ||||
Samuel Clochard | Alexis Hurtrel | ||||
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
44 | ||||||||
| 5. | DECLARATION BY THE PERSON RESPONSIBLE FOR THE INFORMATION | |||||||
“I hereby declare, to the best of my knowledge, that the financial statements for the most recent half year have been prepared in accordance with the applicable accounting standards and give a true and fair view of the assets and liabilities, the financial position and the results of the Company and all the other companies included in the scope of consolidation, and that the half-year management report gives a fair description of the important events of the first six months of the fiscal year and their impact on the half year financial statements, the main related party transactions as well as a description of the main risks and uncertainties for the six months to come.”
Pascal Prigent
Chief Executive Officer
Loos, September 22, 2025
 | Half-Year Business and Financial Report at June 30, 2025 | |||||||
45 | ||||||||
 | ||
| Société anonyme à Conseil d’Administration | ||
au capital social de 12 500 722, 50 euros réparti en 50 002 890 actions de nominal 0,25 euro | ||
| Headquarters: Parc Eurasanté - 885, avenue Eugène Avinée - 59120 LOOS - France | ||
| www.genfit.com | contact@genfit.com | ||
| 424 341 907 R.C.S. Lille Métropole | ||
